The Prostate & Seminal Vesicles
The prostate and seminal vesicles are paired components of the male accessory sex-gland complex, wrapped by a shared investment of pelvic fascia, perfused by a single arterial pedicle, and innervated by the cavernous nerves whose preservation is the central problem of modern pelvic surgery.[5][6][7] For the reconstructive urologist these two organs matter as: (1) the site of the commonest operative disease in men (BPH and prostate cancer), (2) the anatomic neighbors whose dissection determines continence and erectile outcome after prostatectomy, (3) the plane-of-dissection landmark (Denonvilliers') between lower urinary tract and rectum, and (4) the donor of 70%+ of ejaculate, so the target of ejaculation-preserving BPH surgery and of fertility-motivated sparing during radical prostatectomy. This article focuses on the zonal, fascial, vascular, and neural anatomy that drives those operations, and keeps physiology to what is needed to interpret PSA, reason about androgen-dependent disease, and counsel about fertility.
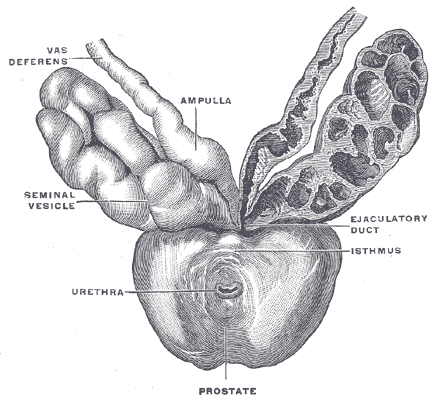 Gray's Anatomy Fig. 1160 — Prostate with seminal vesicles and vas deferens, viewed from in front and above.
Gray's Anatomy Fig. 1160 — Prostate with seminal vesicles and vas deferens, viewed from in front and above.
The Prostate
Gross and zonal anatomy
The prostate is a walnut-sized fibromuscular exocrine gland immediately below the bladder, encircling the prostatic urethra, resting on the urogenital diaphragm, and bounded anteriorly by the pubic symphysis and posteriorly by the rectum (separated by Denonvilliers' fascia). McNeal's zonal anatomy partitions the gland into four regions with distinct disease predispositions and operative relevance:[1][2][3]
| Zone | % of glandular volume | Origin / notes | Disease predilection |
|---|---|---|---|
| Peripheral zone (PZ) | ~70% | Urogenital-sinus origin | ~70% of prostate cancers; target of systematic and MRI-guided biopsy |
| Central zone (CZ) | ~25% | Wolffian duct origin (shares histology/biology with the seminal vesicles)[4] | Relatively cancer-resistant; occasionally invaded by posterior PZ tumors at the base |
| Transition zone (TZ) | Small (enlarges with age) | Periurethral, proximal to verumontanum | Exclusive site of BPH; the TZ expansion is what is enucleated at HoLEP / simple prostatectomy / TURP |
| Anterior fibromuscular stroma (AFS) | — (non-glandular) | Forms the entire anterior surface | Not a cancer site; its thickness must be traversed during retropubic apical dissection |
Surface landmarks used in the operating room:
- Base — superior surface, contiguous with the bladder neck.
- Apex — inferior, continuous with the membranous urethra through the rhabdosphincter.
- Anterior surface — AFS; retropubic / Retzius access.
- Posterior surface — separated from rectum by Denonvilliers' fascia (the key posterior dissection plane in prostatectomy, rectourethral-fistula repair, and pelvic-fracture urethroplasty).
- Lateral surfaces — bracketed by the neurovascular bundles running posterolaterally.
The prostatic urethra and its landmarks
The prostatic urethra runs vertically through the gland (~3–4 cm). The midline verumontanum (colliculus seminalis) is the dominant landmark:
- Utricle — midline Müllerian remnant opening at the verumontanum.
- Ejaculatory ducts — paired openings on either side of the utricle; formed proximally by the junction of the vas deferens (ampulla) and seminal vesicle duct.
- Bladder-neck / 5-α-reductase–responsive proximal urethra — the site of α-adrenergic closure, clinically disrupted by α-blockers (tamsulosin, silodosin), TURP, and bladder-neck incision (BNI) with retrograde-ejaculation as the consequence.
Fascia and the "surgical capsule"
Surrounding the prostate are three layers of pelvic fascia, each relevant to dissection:
- Prostatic capsule — thin fibromuscular layer, densely adherent to the gland.
- Prostatic (visceral) fascia — outer fibrofatty coat carrying the capsular vessels and branches of the pelvic plexus.
- Denonvilliers' fascia — posterior; two layers enveloping the prostate and seminal vesicles and separating them from the anterior rectum.
An additional operative "capsule" is the surgical capsule — the compressed peripheral and central zone tissue outside a hypertrophied transition zone. It is not an anatomic layer but the plane exploited in open simple prostatectomy, HoLEP, and TURP enucleation: the adenoma shells out along this interface because the TZ is histologically and biochemically distinct from the PZ/CZ.
Vascular supply and venous drainage
Arterial supply is dominantly from the prostatic artery, a branch of the inferior vesical artery (from the internal iliac). The prostatic artery bifurcates as it enters the gland:
| Branch | Territory |
|---|---|
| Urethral branches | Enter at the prostatovesical junction to supply the proximal urethra and TZ (the dominant supply to the BPH adenoma — the reason prostatic-artery embolization for BPH targets this branch) |
| Capsular branches | Run posterolaterally within the prostatic fascia, accompanying the cavernous nerves, to supply the PZ and CZ |
Venous drainage coalesces into the periprostatic venous plexus (Santorini's), which communicates anteriorly with the deep dorsal vein of the penis and drains via the internal iliac system to the presacral venous plexus (Batson's). Santorini's plexus is the dominant bleeding source at apical prostatectomy dissection — the dorsal vein complex must be secured before urethral transection.
Lymphatic drainage
Principally to the obturator, internal iliac, and external iliac basins, with secondary drainage to common iliac and presacral nodes. Lymphadenectomy templates in radical prostatectomy reflect this — limited vs extended dissections are defined by these basins.
Innervation and the neurovascular bundles
Autonomic innervation arrives from the inferior hypogastric (pelvic) plexus via two anatomically distinct pathways:[5][6][7][8]
- Posterior (dorsal) route — along the ejaculatory-duct / seminal-vesicle axis to the prostate base.
- Lateral route — along the inferior vesical artery, forming the neurovascular bundle (NVB) on each side of the prostatic fascia.
The NVBs run posterolaterally at approximately the 5 and 7 o'clock positions on either side of the prostate, travel with the prostatic capsular arteries and veins, continue past the apex, and become the cavernous nerves supplying the corpora cavernosa (see The Penis). Autonomic fibers distribute in a spray-like pattern along the posterolateral prostatic surface — they are not a discrete "cord" that can be lifted off; they are a plexus embedded in the periprostatic fascia.[5]
- Athermal apical dissection. Cautery damages the nerves; clips or fine dissection preserve them.
- Plane of dissection. Intrafascial (between the prostatic capsule and the prostatic fascia) preserves the most nerve tissue; interfascial and extrafascial planes progressively sacrifice innervation.
- Both routes must be preserved. Saving only the lateral NVB is insufficient — the posterior route following the seminal vesicle carries an independent contribution. That is why seminal-vesicle-sparing and athermal base dissection matter.[6][8]
- Geometry at 5 and 7 o'clock. Cautery or wide resection at these clock positions is the commonest cause of bilateral nerve injury.
Prostate Physiology — What Matters for the Surgeon
Androgen dependence
Testosterone enters prostate cells and is converted to dihydrotestosterone (DHT) by 5α-reductase type 2, the isozyme dominant in the prostate.[10] DHT binds the androgen receptor (AR) with higher affinity than testosterone and drives epithelial survival and proliferation through both direct epithelial AR signaling and through stromal AR-mediated paracrine signaling (notably via IGF-1).[9]
Clinical translations:
- 5α-reductase inhibitors (finasteride, dutasteride) reduce DHT and shrink the transition zone — the basis of medical BPH therapy. They also lower PSA by roughly half, so interpretation of PSA in patients on these drugs requires doubling the measured value when assessing cancer risk.
- Androgen-deprivation therapy (ADT) — central (LHRH agonists/antagonists) or peripheral (AR antagonists, AR-pathway inhibitors) — is the backbone of treatment for advanced prostate cancer.
- BPH is androgen-dependent; castrate-resistant prostate cancer is not, but usually remains AR-driven.
PSA and the seminal coagulum
PSA (KLK3) is a kallikrein-family serine protease produced by prostatic luminal epithelial cells. Its physiologic function is to liquefy the seminal coagulum by cleaving semenogelin I/II (the scaffold protein secreted by the seminal vesicles).[11] Clinically:
- Serum PSA levels reflect prostatic epithelial mass and the integrity of basement membranes; anything that disrupts glandular architecture (cancer, prostatitis, BPH, instrumentation, biopsy) raises PSA.
- Modifiers of interpretation: age, race, 5α-reductase inhibition (halves PSA), prostate volume (PSA density), DRE/ejaculation-related transient elevations.
Other prostatic secretions
- Citrate — uniquely high in prostatic fluid; zinc-regulated pathway that is disrupted in prostate cancer (MR spectroscopic imaging exploits the resulting choline/citrate ratio).
- Zinc — high concentration; required for seminal function and for the citrate pathway.
- Prostatic acid phosphatase — historic prostate-cancer marker; replaced by PSA.
The Seminal Vesicles
Gross anatomy
Paired accessory glands (~5 cm long; sacculated, resembling a "skein of coral" rather than a single tube)[12] lying on the posterior surface of the bladder, lateral to the ampullae of the vas deferens. Each seminal-vesicle duct joins the ipsilateral vas ampulla to form the ejaculatory duct, which traverses the base of the prostate and opens at the verumontanum.
Key operative relations:
- Superior — ureter (crosses anterior to the seminal vesicle as it enters the trigone — the "water under the bridge" equivalent on the dorsal bladder surface).
- Posterior — rectum, separated by Denonvilliers' fascia.
- Anterior — bladder trigone.
- Lateral — pelvic plexus and posterior fibers of the NVB — the seminal-vesicle tips are the site where the NVB enters the prostate, which is why seminal-vesicle-tip sparing in radical prostatectomy has been proposed to improve erectile outcomes without compromising oncologic margins.
Histology and embryologic origin
Mono-layered columnar secretory epithelium, androgen-dependent and lactoferrin-positive; shares histology and biology with the prostate central zone (both derived from the Wolffian duct) and differs from the PZ/TZ (urogenital-sinus origin).[4] This shared origin is why CZ-invading tumors sometimes extend into seminal vesicles along this axis, and why seminal-vesicle invasion is staged distinctly (pT3b) in prostate cancer.
Secretion and the ejaculatory cascade
Seminal vesicles contribute approximately 70% of ejaculate volume.[13][14] Their secretion supplies:
- Fructose — the dominant energy substrate for sperm metabolism; measurable absence in ejaculate is a marker of ejaculatory-duct obstruction or seminal-vesicle agenesis.
- Semenogelin — the scaffold protein that forms the seminal coagulum immediately after ejaculation; subsequently liquefied by prostatic PSA over 20–30 minutes.[11]
- Prostaglandins — smooth-muscle modulators.
- Alkaline pH buffer, fibronectin, and various immunomodulatory and antimicrobial factors.
Seminal-vesicle secretion is androgen-dependent; castration or AR blockade reduces volume and quality dramatically.[15] Obstruction of the ejaculatory ducts (stones, cysts, inflammatory stricture) produces a low-volume, fructose-negative, acidic ejaculate and is the classical indication for transurethral resection of the ejaculatory ducts (TURED).
Congenital anomalies
- Seminal-vesicle agenesis / hypoplasia — commonly associated with ipsilateral Wolffian-duct anomalies including ipsilateral renal agenesis and with CBAVD / CFTR mutations.
- Zinner syndrome — ipsilateral renal agenesis + ipsilateral seminal-vesicle cyst + ipsilateral ejaculatory-duct obstruction.
- Müllerian-duct cyst and utricular cyst — midline, can present with hematospermia, obstructive symptoms, or recurrent epididymitis.
Clinical Correlations for the Reconstructive Urologist
- BPH surgery plane. The TZ is histologically and biochemically distinct from the PZ/CZ. The "surgical capsule" between them is the plane of HoLEP, open/robotic simple prostatectomy, and TURP enucleation. Recognition of this plane — not a specific technology — is what makes complete adenoma removal possible.
- Ejaculation-preserving BPH surgery. Traditional TURP and bladder-neck incision disrupt the α-adrenergic bladder-neck closure mechanism and produce retrograde ejaculation in a majority of men. Ejaculation-preserving HoLEP / TURP techniques preserve periverumontanal and bladder-neck tissue to maintain antegrade ejaculation; newer minimally invasive therapies (Urolift, Rezum, iTind) are designed around the same principle.
- Prostatic-artery embolization (PAE). Targets the urethral branches of the prostatic artery — the dominant supply to the TZ adenoma — through a femoral or radial catheter approach.
- Radical prostatectomy and nerve-sparing. NVBs sit at 5 and 7 o'clock; athermal dissection, choice of fascial plane (intra- vs inter- vs extrafascial), preservation of apical supports (puboprostatic ligaments, dorsal vein complex), and — when oncologically permissible — seminal-vesicle-tip sparing drive post-operative potency outcomes.
- Continence after prostatectomy. Preservation of membranous urethral length, the rhabdosphincter and its innervation, and the posterior musculofascial support (retrotrigonal plate, Denonvilliers' reconstruction) correlates with earlier return of continence. See Male Urethra for the sphincter-level anatomy.
- Transurethral resection of ejaculatory ducts (TURED). For ejaculatory-duct obstruction causing infertility or pain; cuts through the verumontanum into the dilated ducts posteriorly — complications include retrograde ejaculation, urinary reflux into the ducts (acute epididymitis), and rectal injury.
- Rectourethral fistula. Occurs after prostate-cancer therapy (surgery, radiation, HIFU/cryoablation) and enters the Denonvilliers'-fascia plane. Repair exploits this plane with interposition flaps (gracilis, Martius, dartos) depending on the approach (transperineal, transanal, transabdominal).
- Prostate cancer — where it is, where you biopsy. ~70% of cancers arise in the PZ; MRI-guided and systematic biopsies target this zone preferentially. MpMRI identifies PI-RADS lesions by zone and guides transperineal or transrectal targeting. Seminal-vesicle invasion (pT3b) is an independent adverse feature and influences operative margin strategy and nerve-sparing extent.
- Pelvic fracture urethral injury (PFUI) and perineal prostatectomy / YV-plasty. The prostatic apex sits on the urogenital diaphragm; pelvic-fracture shear disrupts membranous urethra at this junction. Reconstruction requires familiarity with the retropubic fascial planes around the apex.
- Seminal-vesicle pathology and access. Cysts, stones, and abscesses can be approached transperineally, transrectally, or laparoscopically/robotically. The seminal-vesicle tips are the lateral pelvic-plexus neighborhood — dissection must be meticulous.
Videos
References
1. McNeal JE. "The Zonal Anatomy of the Prostate." Prostate. 1981;2(1):35–49. doi:10.1002/pros.2990020105
2. Ali A, Du Feu A, Oliveira P, et al. "Prostate Zones and Cancer: Lost in Transition?" Nat Rev Urol. 2022;19(2):101–115. doi:10.1038/s41585-021-00524-7
3. McNeal JE. "Normal Histology of the Prostate." Am J Surg Pathol. 1988;12(8):619–633. doi:10.1097/00000478-198808000-00003
4. Laczkó I, Hudson DL, Freeman A, Feneley MR, Masters JR. "Comparison of the Zones of the Human Prostate With the Seminal Vesicle: Morphology, Immunohistochemistry, and Cell Kinetics." Prostate. 2005;62(3):260–266. doi:10.1002/pros.20149
5. Mandel A, Parekh S, Choudhary M, et al. "Analysis of the Current Surgical Anatomical Knowledge of Radical Prostatectomy: An Updated Review." Eur Urol. 2025. doi:10.1016/j.eururo.2025.06.002
6. Röthlisberger R, Aurore V, Boemke S, et al. "The Anatomy of the Male Inferior Hypogastric Plexus: What Should We Know for Nerve Sparing Surgery." Clin Anat. 2018;31(6):788–796. doi:10.1002/ca.23079
7. Alanazi G, Alsubaie N, Nabi G, Gillingwater TH, Alashkham A. "Distribution of Neurovascular Structures Within the Prostate Gland and Their Relationship to Complications After Radical Prostatectomy." Prostate. 2024;84(5):491–501. doi:10.1002/pros.24667
8. Alsaid B, Karam I, Bessede T, et al. "Tridimensional Computer-Assisted Anatomic Dissection of Posterolateral Prostatic Neurovascular Bundles." Eur Urol. 2010;58(2):281–287. doi:10.1016/j.eururo.2010.04.002
9. Wen S, Chang HC, Tian J, et al. "Stromal Androgen Receptor Roles in the Development of Normal Prostate, Benign Prostate Hyperplasia, and Prostate Cancer." Am J Pathol. 2015;185(2):293–301. doi:10.1016/j.ajpath.2014.10.012
10. Zhu YS, Imperato-McGinley JL. "5α-Reductase Isozymes and Androgen Actions in the Prostate." Ann N Y Acad Sci. 2009;1155:43–56. doi:10.1111/j.1749-6632.2009.04115.x
11. Lilja H, Oldbring J, Rannevik G, Laurell CB. "Seminal Vesicle-Secreted Proteins and Their Reactions During Gelation and Liquefaction of Human Semen." J Clin Invest. 1987;80(2):281–285. doi:10.1172/JCI113070
12. Lee JS, Yeo IS, Lee HI, et al. "Three-Dimensional Reconstruction of the Luminal Structure of Human Seminal Vesicle." J Anat. 2020;237(6):1006–1014. doi:10.1111/joa.13269
13. Aumüller G, Riva A. "Morphology and Functions of the Human Seminal Vesicle." Andrologia. 1992;24(4):183–196. doi:10.1111/j.1439-0272.1992.tb02636.x
14. Gonzales GF. "Function of Seminal Vesicles and Their Role on Male Fertility." Asian J Androl. 2001;3(4):251–258.
15. Welsh M, Moffat L, Jack L, et al. "Deletion of Androgen Receptor in the Smooth Muscle of the Seminal Vesicles Impairs Secretory Function and Alters Its Responsiveness to Exogenous Testosterone and Estradiol." Endocrinology. 2010;151(7):3374–3385. doi:10.1210/en.2009-1339