The Testicles & Scrotum
The testis is a paired endocrine (testosterone) and exocrine (sperm) organ suspended in the scrotum by the spermatic cord and enveloped in a concentric sleeve of fascia that mirrors the abdominal wall.[1][13] For the reconstructive urologist, these two organs are less about cell biology than about layers, blood supply, and lymphatic drainage: the tunica-vaginalis plane determines hydrocele and hernia repair, the three-source arterial redundancy and the pampiniform plexus geometry determine microsurgical varicocelectomy and orchiopexy, the para-aortic lymphatic drainage determines the inguinal (not scrotal) approach to any testicular mass, and the continuous dartos–Scarpa–Colles' fascial plane explains the characteristic spread of Fournier's gangrene. This article prioritises that surgical anatomy and keeps gametogenic, endocrine, and molecular biology only to the depth a reconstructive urologist needs for counseling about fertility, post-vasectomy behavior, torsion windows, and RPLND templates.
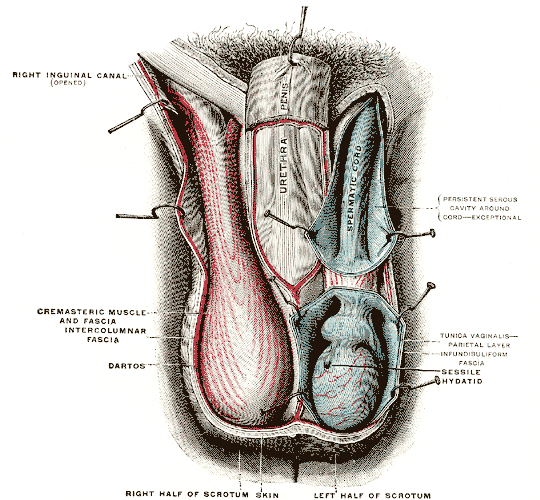 Gray's Anatomy Fig. 1143 — The right testis exposed by laying open the tunica vaginalis.
Gray's Anatomy Fig. 1143 — The right testis exposed by laying open the tunica vaginalis.
Testis — Gross and Surgical Anatomy
The adult testis is a ~4–5 cm ovoid, 15–25 mL organ suspended in the scrotum with its long axis vertical and slightly tilted forward. It is surrounded by a tough fibrous tunica albuginea, which sends septula inward to divide the parenchyma into ~250–300 lobules, each housing 1–4 seminiferous tubules.[2] The tubules converge posteriorly at the mediastinum testis (a thickening of the tunica) to form the rete testis, which drains through ~10–20 efferent ductules into the caput epididymis.
Tunical layers — the operative plane for hydrocele, torsion, and trauma
Surrounding the testis, from deep to superficial:
| Layer | Origin | Operative relevance |
|---|---|---|
| Tunica albuginea | Dense fibrous capsule of the testis itself | The plane of testis-sparing surgery, biopsy, and micro-TESE |
| Tunica vaginalis — visceral layer | Lines the tunica albuginea | The tunica-vaginalis space is where hydroceles accumulate |
| Tunica vaginalis — parietal layer | Lines the inside of the scrotal wall | The closure / resection plane in hydrocele repair (Jaboulay, Lord, or excisional) |
| Internal spermatic fascia | Transversalis fascia | Spermatic-cord investment |
| Cremasteric fascia + cremaster muscle | Internal oblique | Cremasteric reflex; incised during orchiopexy |
| External spermatic fascia | External oblique aponeurosis | Superficial cord investment |
| Dartos fascia | Scarpa's → Colles' fascia continuum | Superficial hematoma / urinoma / Fournier plane; midline septum of the scrotum |
| Scrotal skin | Thin, rugated, hair-bearing | Sweat glands and hair follicles — site of epidermal inclusion cysts, hidradenitis suppurativa[16] |
Spermatic cord contents
Within the cord, from anterior to posterior:
- Testicular artery (from aorta)
- Cremasteric artery (from inferior epigastric)
- Deferential artery (from inferior vesical)
- Pampiniform venous plexus (the three arteries are bracketed by the venous plexus — a critical geometry for microsurgical varicocelectomy)
- Lymphatics to para-aortic nodes
- Vas deferens (posteromedial, palpably firm — the landmark when the surgeon "loses" the cord)
- Genital branch of the genitofemoral nerve (motor to cremaster; sensory to anterior scrotum)
- Ilioinguinal nerve (sensory only; runs on the cord, not inside the internal spermatic fascia)
- Sympathetic and parasympathetic autonomic fibers
Vascular Supply and Drainage
Arterial supply — threefold and redundant
Three paired arteries perfuse the testis and epididymis, with extensive anastomoses along the cord and at the tunica:[18]
| Artery | Origin | Territory |
|---|---|---|
| Testicular artery | Abdominal aorta (below the renal arteries) | Dominant supply; enters the testis at the mediastinum; bifurcates into polar branches |
| Cremasteric artery | Inferior epigastric (external iliac) | Cord, cremaster, and lower-pole anastomosis |
| Deferential artery | Inferior vesical (internal iliac) | Vas deferens and epididymis; anastomoses at the mediastinum |
The cremasteric and deferential supplies are the basis for testicular salvage after testicular-artery injury — the testis can survive ligation of the main testicular artery at the internal ring if the collaterals are intact (a principle exploited in the Fowler–Stephens orchiopexy for high intra-abdominal undescended testes).
Venous drainage — the varicocele anatomy
Veins emerge from the testis at the mediastinum as the pampiniform plexus, a network of 8–12 veins that coalesces as it ascends in the cord to form the testicular (gonadal) vein:[19]
- Left testicular vein → drains into the left renal vein at approximately 90°.
- Right testicular vein → drains directly into the inferior vena cava.
- No valves in the testicular veins; no cross-communication between sides.
Three consequences follow:
- Left-side predominance of varicoceles (85–95%) because of the perpendicular drainage into the left renal vein, the longer left testicular vein column, and the "nutcracker" compression between SMA and aorta.
- Isolated right varicocele mandates imaging to exclude IVC compression or retroperitoneal mass.
- Pampiniform countercurrent heat exchange — the plexus cools inflowing testicular arterial blood from ~37°C to ~33°C, a thermal requirement for normal spermatogenesis. Varicocele disrupts this exchange.
Lymphatic drainage — why orchiectomy is inguinal, not scrotal
Testicular and intratunical lymph drains with the cord upward to the para-aortic / interaortocaval nodes at the L1–L3 level:[20][21][22]
- Right testis → interaortocaval and precaval nodes primarily.
- Left testis → left para-aortic / preaortic nodes primarily.
- Scrotal skin drains to superficial inguinal nodes — a completely separate lymphatic territory.
Any testicular mass suspected of malignancy is removed through an inguinal incision with control at the internal ring, never trans-scrotal. A trans-scrotal approach opens a previously unviolated inguinal lymphatic channel into scrotal skin, potentially upstaging a clinical-stage I germ-cell tumor and broadening the field for adjuvant therapy (inguinal XRT or hemiscrotectomy may be required to salvage).
Innervation
- Autonomic — testicular nerves travel with the testicular artery from the T10–T11 segments via the celiac and intermesenteric plexuses; sympathetic dominance.
- Referred pain from testicular pathology is classically to the flank or ipsilateral lower abdomen (T10–T11 dermatome), paralleling ureteric pain.
- Scrotal sensation — anterior hemiscrotum via genital branch of the genitofemoral nerve (L1–L2) and ilioinguinal nerve (L1); posterior via posterior scrotal nerves from the pudendal (S2–S4). The interscrotal septum is densely innervated, and each hemiscrotum is largely innervated separately — the anatomic basis for unilateral scrotal blocks.[23][24]
- Post-vasectomy pain / chronic orchialgia is thought to involve ilioinguinal, genitofemoral, and spermatic-cord autonomic fibers — microsurgical spermatic-cord denervation selectively transects these while preserving vas, testicular artery, and lymphatics.
Testicular Physiology — Operative Depth Only
Endocrine function
Leydig cells in the interstitium produce ~3–10 mg of testosterone daily under LH drive, with morning peaks in circulating concentration.[4][5] Intratesticular testosterone is 50–100× circulating levels, a detail that matters because:
- Exogenous testosterone suppresses LH/FSH and collapses intratesticular testosterone — the mechanism by which TRT produces oligospermia/azoospermia and the reason fertility counseling must precede its initiation.
- Restoration of spermatogenesis after TRT cessation typically takes 3–6 months; hCG (mimics LH, maintains intratesticular testosterone) and SERMs (clomiphene, enclomiphene) are used for fertility-preserving testosterone optimization.
Spermatogenesis — counseling numbers
Spermatogenesis takes approximately 74 days from spermatogonial stem cell to mature spermatozoon in the tubular lumen, with an additional 10–14 days of epididymal transit and maturation.[6][7] Daily sperm output is ~45 million per testis (~1,000 per second).[6] Clinical translations:
- Post-vasectomy semen analysis is typically performed 8–12 weeks and ~20 ejaculations after surgery, reflecting the time to clear stored sperm distal to the vasectomy site.
- Gonadotoxic exposures (chemotherapy, radiation) damage the stem / differentiating pool; recovery timelines parallel the 74-day cycle plus stem-cell regeneration.
- Fertility preservation (sperm banking) is the standard of care before any gonadotoxic therapy.
Blood–testis barrier
Sertoli–Sertoli tight junctions create a blood–testis barrier (BTB) separating a basal (spermatogonial) from an adluminal (spermatocyte and spermatid) compartment. The BTB is an immune privilege site; its disruption (vasectomy, trauma, torsion) can produce anti-sperm antibodies that compromise subsequent fertility and are a factor in vasovasostomy outcome.[3]
Thermoregulation
Normal testicular temperature is ~33°C, 2–4°C below core temperature. Four mechanisms cooperate:[12][14][15][17]
- Pampiniform plexus — countercurrent heat exchange (the main mechanism).
- Cremaster muscle — retracts testis toward body in cold.
- Dartos muscle — wrinkles and shortens the scrotal skin in cold; thins and elongates in heat.
- Scrotal sweating — thin skin with dense sweat glands dissipates heat.
Elevated scrotal temperature (varicocele, cryptorchidism, chronic heat exposure, sedentary position) impairs spermatogenesis by inducing germ-cell apoptosis and BTB disruption — the pathophysiologic rationale for varicocelectomy in infertility.
The Epididymis and Vas Deferens
The epididymis runs along the posterolateral surface of the testis and has three operatively useful regions:[8][9]
| Segment | Function | Clinical corollary |
|---|---|---|
| Caput (head) | Receives sperm from efferent ductules; initial maturation | Site of efferent-ductule–rete-testis obstruction (CBAVD, post-inflammatory); target of MESA and PESA sperm retrieval |
| Corpus (body) | Primary maturation zone; sperm acquire progressive motility and fertilizing capacity | Site of post-infectious (chlamydia, gonorrhea, tuberculosis) stricture |
| Cauda (tail) | Sperm storage in quiescent state | Source of stored ejaculated sperm; the site where vasectomy leaves residual sperm for weeks after |
The epididymis terminates as the vas deferens, which ascends through the cord, enters the internal ring, and runs retroperitoneally to the bladder base, joining the seminal-vesicle duct to form the ejaculatory duct (see The Prostate & Seminal Vesicles).
Epididymal sperm maturation is androgen-dependent, proceeds without sperm gene transcription, and is mediated by segment-specific secretions and epididymosomes (epithelial exosomes delivering proteins, lipids, and miRNAs to spermatozoa).[10][11] This is why obstructive azoospermia upstream of the cauda yields immature, poorly motile sperm — a fact reflected in retrieval strategy (testicular > epididymal sperm in long-standing obstruction; vice versa in recent obstruction).
The Scrotum
Layers (superficial → deep) — the abdominal-wall homologues
| Scrotal layer | Abdominal-wall homologue | Operative note |
|---|---|---|
| Skin | Skin | Thin, no subcutaneous fat, rugated under dartos tone |
| Dartos fascia + dartos muscle | Scarpa's fascia / Colles' fascia | Continuous with the perineum and anterior abdominal wall — the fascial highway of Fournier's gangrene spread |
| External spermatic fascia | External oblique aponeurosis | |
| Cremasteric fascia + muscle | Internal oblique | Cremasteric reflex (L1–L2 genitofemoral) |
| Internal spermatic fascia | Transversalis fascia | Deepest investment of the cord |
| Tunica vaginalis (parietal) | Parietal peritoneum (processus vaginalis) | Site of hydrocele; communicating hydrocele = patent processus vaginalis |
Arterial supply
- Anterior hemiscrotum — branches of the external pudendal arteries (from the femoral)
- Posterior hemiscrotum — branches of the internal pudendal arteries (from the internal iliac)
This split supports scrotal flap design for reconstruction and explains the robustness of scrotal viability even with extensive debridement.
Dartos-fascia continuity and Fournier's gangrene
Dartos fascia is continuous superiorly with Scarpa's fascia of the anterior abdominal wall and posteriorly with Colles' (perineal superficial) fascia. Fournier's gangrene respects these fascial boundaries and spreads from the perineum onto the anterior abdominal wall and scrotum along this continuous plane — while the deep cremasteric/internal spermatic layers, and the testis itself (separately supplied and covered), are typically spared. That is why the testes and cord are preserved during Fournier's debridement whenever possible.
Clinical Correlations for the Reconstructive Urologist
- Testicular torsion. A surgical emergency. Bell-clapper deformity (complete investment of the testis by the tunica vaginalis, "clapper in a bell") is the anatomic substrate and is typically bilateral — contralateral orchiopexy at the same operation is standard. Salvage depends on duration of ischemia (~100% at <6 h, declining rapidly thereafter). Detorsion direction is "out and lateral" (opening a book from the midline).
- Cryptorchidism and orchiopexy. Most common male birth defect; long-term risks are infertility and testicular germ-cell tumor (RR ~2–8). Inguinal canalicular testis → standard inguinal orchiopexy. Intra-abdominal testis with insufficient cord length → Fowler–Stephens staged orchiopexy, relying on the deferential and cremasteric collaterals after sacrifice of the testicular artery.
- Hydrocele and hernia. Communicating hydrocele / indirect inguinal hernia reflect a patent processus vaginalis. Non-communicating adult hydrocele is managed by Jaboulay (eversion), Lord (plication), or excisional technique, addressing the tunica-vaginalis parietal layer.
- Varicocele. Dilation of the pampiniform plexus, ~85–95% left-sided. Isolated right varicocele or acute/new varicocele warrants imaging for IVC obstruction. Grading (I–III) and surgical indication rest on fertility, pain, and atrophy. Microsurgical subinguinal varicocelectomy is the gold standard — preserves testicular arteries and lymphatics, ligates internal-spermatic and external-spermatic veins plus gubernacular collaterals.
- Testicular germ-cell tumor. Arises from germ-cell neoplasia in situ (GCNIS). Radical inguinal orchiectomy with control at the internal ring; trans-scrotal approach is contraindicated. Para-aortic/interaortocaval nodes are the first landing zone — the anatomic basis of RPLND templates (modified templates preserve sympathetic chains L1–L3 and thus antegrade ejaculation).
- Fournier's gangrene. Necrotizing fasciitis of the dartos / Scarpa's / Colles' plane. Emergent wide debridement with urinary and fecal diversion as needed; testes, cord, and glans penis usually preserved. Reconstruction later with split-thickness grafts, fasciocutaneous flaps, or muscle flaps (gracilis) depending on defect.
- Sperm retrieval. MESA / PESA (obstructive azoospermia, proximal ducts dilated with mature sperm), TESE (non-obstructive azoospermia, focal spermatogenesis), micro-TESE (operating microscope to identify dilated opaque seminiferous tubules containing active spermatogenesis — the procedure of choice for non-obstructive azoospermia).
- Vasectomy and vasoreversal. Technique: isolation, ligation, fascial interposition. Post-vasectomy semen analysis at ~3 months / ≥20 ejaculations. Vasovasostomy success is strongly time-dependent (>95% patency at <3 years, declining); vasoepididymostomy is required when obstructive interval is long and epididymal sperm granulomas have caused secondary obstruction — the longer the interval, the higher the likelihood a vasoepididymostomy will be required.
- Testicular prosthesis. Offered at the time of orchiectomy or later; silicone implant in the scrotal compartment. Counsel about size options, palpation, and the (small) rate of infection / extrusion.
- Scrotal reconstruction. After Fournier's, penile cancer en bloc, or burn / avulsion. Options progress from primary closure / split-thickness graft → local fasciocutaneous flaps → musculocutaneous (gracilis, medial thigh). The testes can be temporized in medial-thigh pouches if the scrotum is not immediately reconstructable.
- Chronic orchialgia. Medical management first (NSAIDs, neuropathic agents, pelvic-floor PT). Diagnostic spermatic-cord block is the gateway to microsurgical spermatic-cord denervation, which ligates cord autonomic fibers while preserving vas, testicular and deferential arteries, and lymphatics.
- Referred pain — the crossovers. Testicular pathology refers to the flank via T10–T11; ureteric pathology refers to the scrotum / groin via the same dermatomes. A patient with acute flank and testicular pain needs both ureter and testis evaluated.
References
1. Mäkelä JA, Koskenniemi JJ, Virtanen HE, Toppari J. "Testis Development." Endocr Rev. 2019;40(4):857–905. doi:10.1210/er.2018-00140
2. Trainer TD. "Histology of the Normal Testis." Am J Surg Pathol. 1987;11(10):797–809. doi:10.1097/00000478-198710000-00007
3. Mruk DD, Cheng CY. "The Mammalian Blood-Testis Barrier: Its Biology and Regulation." Endocr Rev. 2015;36(5):564–591. doi:10.1210/er.2014-1101
4. Zirkin BR, Papadopoulos V. "Leydig Cells: Formation, Function, and Regulation." Biol Reprod. 2018;99(1):101–111. doi:10.1093/biolre/ioy059
5. Basaria S. "Male Hypogonadism." Lancet. 2014;383(9924):1250–1263. doi:10.1016/S0140-6736(13)61126-5
6. Griswold MD. "Spermatogenesis: The Commitment to Meiosis." Physiol Rev. 2016;96(1):1–17. doi:10.1152/physrev.00013.2015
7. Nishimura H, L'Hernault SW. "Spermatogenesis." Curr Biol. 2017;27(18):R988–R994. doi:10.1016/j.cub.2017.07.067
8. Zhou W, De Iuliis GN, Dun MD, Nixon B. "Characteristics of the Epididymal Luminal Environment Responsible for Sperm Maturation and Storage." Front Endocrinol. 2018;9:59. doi:10.3389/fendo.2018.00059
9. Sipilä P, Björkgren I. "Segment-Specific Regulation of Epididymal Gene Expression." Reproduction. 2016;152(3):R91–R99. doi:10.1530/REP-15-0533
10. Ali W, Deng K, Bian Y, Liu Z, Zou H. "Spectacular Role of Epididymis and Bio-Active Cargo of Nano-Scale Exosome in Sperm Maturation." Biomed Pharmacother. 2023;164:114889. doi:10.1016/j.biopha.2023.114889
11. Chen H, Alves MBR, Belleannée C. "Contribution of Epididymal Epithelial Cell Functions to Sperm Epigenetic Changes and the Health of Progeny." Hum Reprod Update. 2021;28(1):51–66. doi:10.1093/humupd/dmab029
12. Skandhan KP, Rajahariprasad A. "The Process of Spermatogenesis Liberates Significant Heat and the Scrotum Has a Role in Body Thermoregulation." Med Hypotheses. 2007;68(2):303–307. doi:10.1016/j.mehy.2006.06.058
13. Raad G, Massaad V, Serdarogullari M, et al. "Functional Histology of Human Scrotal Wall Layers and Their Overlooked Relation With Infertility: A Narrative Review." Int J Impot Res. 2023;35(5):428–438. doi:10.1038/s41443-022-00573-5
14. Waites GM. "Thermoregulation of the Scrotum and Testis: Studies in Animals and Significance for Man." Adv Exp Med Biol. 1991;286:9–17. doi:10.1007/978-1-4684-5913-5_2
15. Ivell R. "Lifestyle Impact and the Biology of the Human Scrotum." Reprod Biol Endocrinol. 2007;5:15. doi:10.1186/1477-7827-5-15
16. Hamad J, McCormick BJ, Sayed CJ, et al. "Multidisciplinary Update on Genital Hidradenitis Suppurativa: A Review." JAMA Surg. 2020;155(10):970–977. doi:10.1001/jamasurg.2020.2611
17. Sealfon AI, Zorgniotti AW. "A Theoretical Model for Testis Thermoregulation." Adv Exp Med Biol. 1991;286:123–135. doi:10.1007/978-1-4684-5913-5_12
18. Mostafa T, Labib I, El-Khayat Y, El-Rahman El-Shahat A, Gadallah A. "Human Testicular Arterial Supply: Gross Anatomy, Corrosion Cast, and Radiologic Study." Fertil Steril. 2008;90(6):2226–2230. doi:10.1016/j.fertnstert.2007.10.013
19. Wishahi MM. "Anatomy of the Venous Drainage of the Human Testis: Testicular Vein Cast, Microdissection and Radiographic Demonstration." Eur Urol. 1991;20(2):154–160. doi:10.1159/000471687
20. Oka S, Shiraishi K, Matsuyama H. "Microsurgical Anatomy of the Spermatic Cord and Spermatic Fascia: Distribution of Lymphatics, and Sensory and Autonomic Nerves." J Urol. 2016;195(6):1841–1847. doi:10.1016/j.juro.2015.11.041
21. Holstein AF, Orlandini GE, Möller R. "Distribution and Fine Structure of the Lymphatic System in the Human Testis." Cell Tissue Res. 1979;200(1):15–27. doi:10.1007/BF00236883
22. Yeh SD, Morse MJ, Grando R, Kleinert EL, Whitmore WF. "Lymphoscintigraphic Studies of Lymphatic Drainage From the Testes." Clin Nucl Med. 1986;11(12):823–827. doi:10.1097/00003072-198612000-00004
23. Amid PK. "A 1-Stage Surgical Treatment for Postherniorrhaphy Neuropathic Pain: Triple Neurectomy and Proximal End Implantation Without Mobilization of the Cord." Arch Surg. 2002;137(1):100–104. doi:10.1001/archsurg.137.1.100
24. Yucel S, Baskin LS. "The Neuroanatomy of the Human Scrotum: Surgical Ramifications." BJU Int. 2003;91(4):393–397. doi:10.1046/j.1464-410x.2003.04087.x