The Uterus
The uterus is a muscular organ whose function — implantation, gestation, parturition, and menstrual shedding — is almost entirely reproductive. For the reconstructive pelvic surgeon, however, it matters as a structural and anatomical entity rather than as an endocrine or obstetric one: the uterus anchors the Level-I apical support through the cardinal and uterosacral complex, shares a posterolateral vascular–neural pedicle with the cervix that contains the ureter and pelvic plexus, and is the operative neighbor whose manipulation (hysterectomy, myomectomy, cesarean section, arterial embolization) generates a large fraction of the urogynecologic workload — from vesicovaginal fistula to apical vault prolapse to radiation and surgical neurogenic bladder. This article concentrates on the surgical anatomy, ligamentous support, vascular pedicle, and innervation that matter for the reconstructive surgeon, and condenses menstrual-cycle endocrinology and molecular parturition biology to the minimum useful depth.
See also The Cervix (contiguous organ sharing most of these supports); The Vagina for DeLancey's support model; and The Ureters for the "water under the bridge" anatomy at the cardinal ligament.
 Gray's Anatomy Fig. 1170 — The uterus and right broad ligament, seen from behind.
Gray's Anatomy Fig. 1170 — The uterus and right broad ligament, seen from behind.
Gross Anatomy
The uterus is a pear-shaped fibromuscular organ sitting between bladder (anterior) and rectum (posterior), suspended in the pelvis by four paired ligamentous attachments and the peritoneal broad ligament. Typical dimensions in nulliparous reproductive-age women: 7–8 cm long, 4–5 cm wide, 2–3 cm thick, with substantial variation by age and parity.[5][6][7] Parous uterine volume is ~40% greater than nulliparous.
Three anatomic regions:
| Region | Location | Operative relevance |
|---|---|---|
| Fundus | Above the insertion of the fallopian tubes | Palpable landmark at delivery and during laparoscopy; cornual-region pregnancy is classically "interstitial" here |
| Corpus (body) | Main muscular mass | Site of fibroids, adenomyosis, endometrial cancer; the operative field of myomectomy and hysterectomy |
| Cervix | See The Cervix | Level-I support anchor; ureteric neighbor |
The junction between corpus and cervix (the isthmus) is anatomically where the uterus narrows and where cesarean hysterotomy is performed (low transverse), where cesarean scar defects (isthmocele) form, and where supracervical hysterectomy transects the uterus.
Position and relations
- Anteverted and anteflexed in ~75–80% of women (angle of the uterus relative to the cervix, and of the cervix relative to the vagina, both forward)
- Retroverted / retroflexed in ~20% — a normal variant, but relevant to pessary fit, IUD placement, and surgical orientation
- Anterior: bladder, via the vesicouterine pouch (anterior cul-de-sac)
- Posterior: rectum, via the rectouterine pouch of Douglas (posterior cul-de-sac) — deepest peritoneal recess in the pelvis, the site of enterocele and of posterior culdotomy access
- Lateral: broad ligament with its contents (see below); pelvic sidewall further laterally
Wall Structure
Three layers, with a clinically distinct junctional zone visible on MRI:[3][4][5]
| Layer | Composition | Operative relevance |
|---|---|---|
| Endometrium | Functionalis (proliferates and sheds) + basalis (regenerative) | Site of cyclic bleeding, endometrial hyperplasia and cancer, menstrual dysfunction — the target of endometrial ablation; sampling via pipelle / D&C |
| Junctional zone | Inner myometrial band with tightly packed smooth muscle; MRI-visible | Thickening (>12 mm) is the imaging signature of adenomyosis; thickening peaks at 41–50 years and retreats post-menopause |
| Myometrium | >95% smooth muscle in interlacing bundles; intrinsic pacemaker activity | Site of leiomyomas (submucosal / intramural / subserosal); the dissected territory of myomectomy; the contracting organ at labor and in postpartum hemorrhage |
| Perimetrium (serosa) | Peritoneal covering on most of the outer surface; continuous with broad ligament laterally | The serosal plane of adhesiolysis; absent over the lower anterior segment where the bladder reflects, which is why cesarean and hysterectomy bladder-peritoneal dissection is done in this avascular plane |
Ligamentous Support — The Apical Framework
The uterus is "suspended in air" by four paired attachments, not all of which are true ligaments. For the reconstructive surgeon only two are load-bearing; the others function as conduits or positional tethers.[18][19][20][21][22]
| Structure | Function | Contents |
|---|---|---|
| Cardinal ligament (Mackenrodt / transverse cervical) | Primary load-bearing Level-I support; suspends the cervix/upper vagina from the pelvic sidewall | Uterine artery and veins; ureter at its base; pelvic plexus autonomic fibers; lymphatics |
| Uterosacral ligament | Posterior Level-I support; also the site of USL suspension procedures | Sympathetic fibers of the pelvic plexus, lymphatics |
| Round ligament | Positional only — maintains anteversion; not load-bearing | From uterine cornua → inguinal canal → labia majora; contains Sampson's artery (branch of uterine or ovarian) |
| Broad ligament | Peritoneal drape; carries vessels and tubes | Two layers of peritoneum enclosing fallopian tubes, ovarian ligaments, round ligaments, and the uterine vessels laterally |
Cadaveric and MRI studies demonstrate that the cardinal ligament is not a discrete fibrous band but a perivascular/perineural sheath — essentially a visceral mesentery of vessels, lymphatics, autonomic nerves, and fatty connective tissue extending between the pelvic sidewall and the cervix. This is clinically important: radical-hysterectomy nomenclature (Piver / Querleu-Morrow) is about how laterally in this sheath the dissection extends, which determines how much of the pelvic plexus is sacrificed.[19]
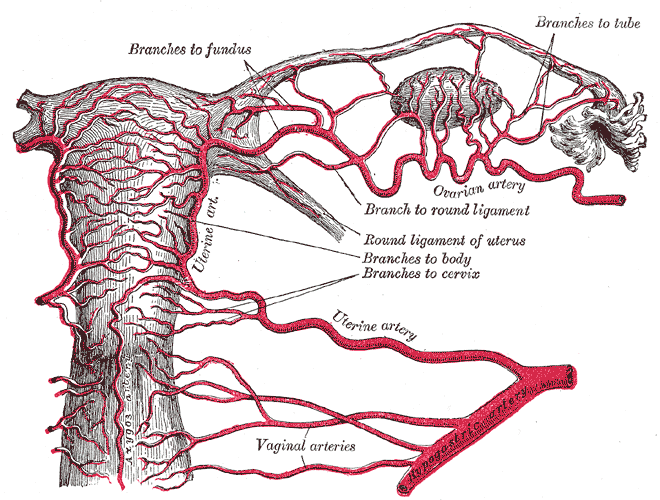
Vascular Supply
Arterial supply is dominantly from the uterine artery (a branch of the internal iliac anterior division), with ovarian-artery anastomoses providing a second axis of supply that is clinically exploited and sometimes clinically surprising:[12][13][14][15]
| Source | Course | Territory |
|---|---|---|
| Uterine artery | Internal iliac → crosses over the ureter 1.5–2 cm lateral to the cervix → ascends in the broad ligament along the lateral uterus | Dominant supply; gives arcuate → radial → spiral arteries into endometrium |
| Ovarian artery | From aorta → infundibulopelvic (suspensory) ligament → ovary → anastomoses with the uterine artery at the cornua | Upper uterus, fundus, tubes |
| Contralateral uterine | Across-midline anastomoses with opposite uterine artery | Collateral supply; why unilateral uterine-artery ligation does not devascularize the uterus |
Ovarian-uterine territorial shift — functional, not fixed
The border between uterine and ovarian arterial territories shifts dynamically across the menstrual cycle: in the follicular phase the dominant-follicle side draws more perfusion from the ovarian artery; in the luteal phase perfusion shifts back to the uterine artery.[12][15] Clinical implication for the reconstructive / interventional surgeon: uterine-artery embolization (UAE) for fibroids or postpartum hemorrhage may fail when ovarian-artery anastomoses are dominant — preprocedural imaging and intraprocedural awareness of these collaterals determine technical success.
Veno-arterial countercurrent exchange
Uterine veins drain to the internal iliac venous plexus; some join the ovarian venous outlet. Close apposition of veins and arteries in the broad ligament supports countercurrent transfer of locally-released substances (estrogen, progesterone, prostaglandins) between ovary, tube, and uterus on the ipsilateral side — which is the pharmacokinetic basis for the vaginally-applied progesterone / hCG producing high uterine concentrations through local rather than systemic transfer.[12]
Innervation
Autonomic, with sparse somatic contribution:[11][16][17]
- Sympathetic — T10–L2 → superior hypogastric plexus (at the sacral promontory) → paired hypogastric nerves → inferior hypogastric (pelvic) plexus → uterus via the cardinal and uterosacral ligaments. Mediates uterine smooth-muscle contraction and vasoconstriction.
- Parasympathetic — S2–S4 pelvic splanchnic nerves → pelvic plexus → uterus via the same ligamentous routes. Vasodilation and relaxation.
- Afferent pain — visceral afferents track with the sympathetic input; labor pain below the cervix is parasympathetic-S2-S4, above via T10–L2 — the distribution targeted by neuraxial labor analgesia and by superior hypogastric plexus block.
The pelvic plexus sits between the cardinal ligament ventrally and the uterosacral ligament dorsally. Preserving bladder-, rectum-, and vaginal-sexual function during radical hysterectomy turns on sparing the posterolateral component of this plexus — see The Cervix for the nerve-sparing radical-hysterectomy details.
Physiology (kept brief)
Menstrual cycle — the endometrial framework
The ~28-day cycle under the hypothalamic–pituitary–ovarian axis produces three functional phases:[2][8][9][10]
- Menstrual (days 1–5) — progesterone withdrawal → spiral-artery vasoconstriction → ischemic shedding of the functionalis.
- Proliferative (days 6–14, estrogen-dominant) — basalis-driven regeneration, gland formation, stromal proliferation, spiral-artery growth. Endometrial thickness rises from 2–4 mm to 8–12 mm.
- Secretory (days 15–28, progesterone-dominant) — glandular coiling and secretion, stromal decidualization, and the window of implantation (days 20–24).
Anomalous endometrial thickness, abnormal uterine bleeding, and the behavior of the endometrium in postmenopausal women (any thickness >4 mm with bleeding warrants sampling) are the practical imaging/clinic translations of this cycle.
Myometrial quiescence and labor
During pregnancy the myometrium is held quiescent by progesterone-dominant signaling (PR suppression of inflammatory transcription factors, repression of contraction-associated proteins, potassium-channel-mediated hyperpolarization). At labor, functional progesterone withdrawal (decline in PR activity rather than in circulating progesterone in humans) lifts this suppression: oxytocin-receptor, connexin-43, and COX-2 expression rise, calcium-channel activity rises, and coordinated contraction becomes possible.[1][23][24][25][26]
Clinical translations for the reconstructive surgeon: oxytocin and prostaglandins are uterotonics for postpartum hemorrhage; β-mimetics and nifedipine are tocolytics; and uterine atonic hemorrhage is managed in a stepwise fashion (uterotonics → intrauterine tamponade [Bakri balloon] → compression sutures [B-Lynch] → uterine / internal iliac artery ligation → emergent hysterectomy).
Pregnancy remodeling
The uterus undergoes massive smooth-muscle hypertrophy and hyperplasia, dramatic vascular expansion, and decidualization of the stroma. It returns toward baseline over the puerperium through apoptosis and matrix remodeling — the same pathway that, when disordered, produces placental invasion anomalies (accreta / increta / percreta).
Clinical Correlations for the Reconstructive Surgeon
- Hysterectomy — approach selection. Vaginal > laparoscopic / robotic > abdominal, in that order of preference for benign disease when anatomy allows, per modern guidelines. The uterine-artery pedicle sits lateral to the cervix with the ureter immediately inferior (see The Cervix); identification of the ureter before clamping is the primary injury-prevention step.
- Ureteric injury. The most common iatrogenic ureteric injury is at the uterine-artery crossing. Risk is concentrated in radical hysterectomy, deep paravaginal dissection, and management of post-cesarean hematoma. See The Ureters.
- Supracervical (subtotal) hysterectomy. Preserves the cervix and its ligamentous attachments. Advantages: faster, potentially fewer urinary and sexual complaints; disadvantages: patient must continue cervical-cancer screening, and cyclical "spotting" from residual endocervical tissue can persist.
- Myomectomy and fibroid mapping. Fibroids are mapped as submucosal (FIGO type 0–2), intramural (3–4), subserosal (5–7), or other (8 — e.g. cervical, parasitic). Submucosal fibroids are removed hysteroscopically; the rest are approached laparoscopically or openly depending on size, number, and fertility plans. The reconstructive surgeon should understand this language because myomectomy scar is a risk factor for rupture in subsequent pregnancy and for intraoperative findings of dense pelvic adhesions at later urogynecologic operation.
- Uterine artery embolization (UAE). Useful for symptomatic fibroids, adenomyosis, AVMs, and postpartum hemorrhage. Technical failure is most often due to dominant ovarian-artery collateral supply — a consideration in preprocedural counseling and imaging. UAE reduces fertility and is not recommended in women with active fertility plans.
- Adenomyosis. Ectopic endometrium within the myometrium → chronic pelvic pain, dysmenorrhea, heavy bleeding. Diagnosed by junctional-zone thickening >12 mm on MRI. Management: hormonal suppression, UAE, focal resection, or hysterectomy.
- Cesarean section and its downstream reconstruction. C-section is the leading cause of iatrogenic bladder injury and vesicovaginal fistula worldwide. Placenta accreta spectrum (accreta / increta / percreta), now increasing with repeat C-sections, often requires emergent cesarean hysterectomy with multidisciplinary urology support for ureteric stenting, bladder repair, and massive hemorrhage. Cesarean scar defect (isthmocele) at the low-transverse scar can cause postmenstrual spotting, pelvic pain, and secondary infertility, and is reconstructed hysteroscopically or laparoscopically.
- Obstetric fistula. Obstructed labor → pressure necrosis of the bladder / rectum against the pubis → vesicovaginal / rectovaginal fistula weeks later. Globally the single most common cause of VVF; in high-resource settings, post-hysterectomy fistula dominates. Treatment: staged transvaginal or abdominal repair with interposition flap; see The Vagina.
- Apical prolapse with uterine descent. Options preserve or remove the uterus: vaginal hysterectomy with USLS or SSLF, sacrospinous hysteropexy, sacrohysteropexy, or Manchester (Fothergill) procedure (cervical amputation + CUL plication). Patient counseling turns on fertility plans, cervix-cancer risk, and prior operations.
- Postoperative bladder, bowel, and sexual dysfunction. Dysfunction after hysterectomy is typically neural (pelvic-plexus injury at the cardinal/uterosacral region) rather than structural. Minimizing this is the core of nerve-sparing technique.
- Uterine rupture. Usually at the site of a prior scar (C-section, myomectomy). Surgical emergency with fetal and maternal risk.
- Placental anomalies and emergent hysterectomy. The reconstructive urologist is increasingly part of the accreta team — preoperative ureteric stenting, intraoperative bladder / ureter dissection, postoperative continence assessment. Knowing the uterine-artery–ureter geometry is non-negotiable.
- Endometrial cancer. Most common gynecologic malignancy in high-resource settings. Staging hysterectomy + bilateral salpingo-oophorectomy ± lymphadenectomy. Radiation for higher stage or recurrence produces radiation cystitis, fistula, ureteric stricture, and bladder atony — the familiar reconstructive late-morbidity profile.
- Gender-affirming hysterectomy. Standard component of masculinizing surgery for gender-diverse patients; usually laparoscopic or robotic total hysterectomy with bilateral salpingectomy ± oophorectomy. Technical considerations are standard; pre-procedural imaging reviews Müllerian anomalies and the effect of long-term testosterone on the endometrium.
- Müllerian anomalies. Septate, bicornuate, didelphys, unicornuate uterus, and MRKH (Müllerian agenesis) have implications for fertility, obstetric outcomes, and reconstructive options (hysteroscopic septal resection, neovagina creation). Cross-link to The Vagina for neovaginal options in MRKH.
- Uterine transplantation. Investigational-to-emerging option for uterine-factor infertility (MRKH, prior hysterectomy). Technically complex vascular anastomoses; urologists are typically involved for ureteric planning and surveillance.
- IUD perforation. Uncommon but clinically important — can migrate to bladder, bowel, or peritoneum. Retrieval is hysteroscopic, laparoscopic, or open depending on location.
References
1. Smith R. "Parturition." N Engl J Med. 2007;356(3):271–283. doi:10.1056/NEJMra061360
2. Borzutzky C, Jaffray J. "Diagnosis and Management of Heavy Menstrual Bleeding and Bleeding Disorders in Adolescents." JAMA Pediatr. 2020;174(2):186–194. doi:10.1001/jamapediatrics.2019.5040
3. Kuperman T, Gavriel M, Gotlib R, et al. "Tissue-Engineered Multi-Cellular Models of the Uterine Wall." Biomech Model Mechanobiol. 2020;19(5):1629–1639. doi:10.1007/s10237-020-01296-6
4. Maruyama T, Masuda H, Ono M, Kajitani T, Yoshimura Y. "Human Uterine Stem/Progenitor Cells: Their Possible Role in Uterine Physiology and Pathology." Reproduction. 2010;140(1):11–22. doi:10.1530/REP-09-0438
5. Hauth EA, Jaeger HJ, Libera H, Lange S, Forsting M. "MR Imaging of the Uterus and Cervix in Healthy Women: Determination of Normal Values." Eur Radiol. 2007;17(3):734–742. doi:10.1007/s00330-006-0313-3
6. Gergolet M, Nicolì P, Bokal EV, et al. "Defining the 'Normal Uterus' by Ultrasound Measurement of Uterine Lengths, Thicknesses, and Angles in a Population of Nulliparous Women: The Normal UteRus asSEssment (NURSE) Study." Fertil Steril. 2025. doi:10.1016/j.fertnstert.2025.07.1220
7. Chen F, Gong Y, Xie Y, et al. "Assessment of Key Parameters of Normal Uterus in Women of Reproductive Age." Sci Rep. 2023;13(1):17794. doi:10.1038/s41598-023-44489-6
8. Critchley HOD, Maybin JA, Armstrong GM, Williams ARW. "Physiology of the Endometrium and Regulation of Menstruation." Physiol Rev. 2020;100(3):1149–1179. doi:10.1152/physrev.00031.2019
9. Wang W, Vilella F, Alama P, et al. "Single-Cell Transcriptomic Atlas of the Human Endometrium During the Menstrual Cycle." Nat Med. 2020;26(10):1644–1653. doi:10.1038/s41591-020-1040-z
10. "Practice Bulletin No. 136: Management of Abnormal Uterine Bleeding Associated With Ovulatory Dysfunction." Obstet Gynecol. 2013;122(1):176–185. doi:10.1097/01.AOG.0000431815.52679.bb
11. Wray S, Prendergast C. "The Myometrium: From Excitation to Contractions and Labour." Adv Exp Med Biol. 2019;1124:233–263. doi:10.1007/978-981-13-5895-1_10
12. Cicinelli E, Einer-Jensen N, Galantino P, Alfonso R, Nicoletti R. "The Vascular Cast of the Human Uterus: From Anatomy to Physiology." Ann N Y Acad Sci. 2004;1034:19–26. doi:10.1196/annals.1335.002
13. Höckel M, Horn LC, Fritsch H. "Association Between the Mesenchymal Compartment of Uterovaginal Organogenesis and Local Tumour Spread in Stage IB-IIB Cervical Carcinoma." Lancet Oncol. 2005;6(10):751–756. doi:10.1016/S1470-2045(05)70324-7
14. Kristek J, Kachlik D, Sticova E, Fronek J. "Contralateral and Ipsilateral Arterial Vasculature of the Human Uterus: The Pilot Results of an Anatomical Study." Physiol Res. 2022;71(Suppl 1):S89–S97.
15. Cicinelli E, Einer-Jensen N, Barba B, et al. "Blood to the Cornual Area of the Uterus Is Mainly Supplied From the Ovarian Artery in the Follicular Phase and From the Uterine Artery in the Luteal Phase." Hum Reprod. 2004;19(4):1003–1008. doi:10.1093/humrep/deh171
16. Di Tommaso S, Cavallotti C, Malvasi A, et al. "A Qualitative and Quantitative Study of the Innervation of the Human Non Pregnant Uterus." Curr Protein Pept Sci. 2017;18(2):140–148. doi:10.2174/1389203717666160330105341
17. Kietpeerakool C, Aue-Aungkul A, Galaal K, Ngamjarus C, Lumbiganon P. "Nerve-Sparing Radical Hysterectomy Compared to Standard Radical Hysterectomy for Women With Early Stage Cervical Cancer (Stage Ia2 to IIa)." Cochrane Database Syst Rev. 2019;2:CD012828. doi:10.1002/14651858.CD012828.pub2
18. Kaniewska M, Gołofit P, Heubner M, Maake C, Kubik-Huch RA. "Suspensory Ligaments of the Female Genital Organs: MRI Evaluation With Intraoperative Correlation." Radiographics. 2018;38(7):2195–2211. doi:10.1148/rg.2018180089
19. Ramanah R, Berger MB, Parratte BM, DeLancey JO. "Anatomy and Histology of Apical Support: A Literature Review Concerning Cardinal and Uterosacral Ligaments." Int Urogynecol J. 2012;23(11):1483–1494. doi:10.1007/s00192-012-1819-7
20. Kieserman-Shmokler C, Swenson CW, Chen L, et al. "From Molecular to Macro: The Key Role of the Apical Ligaments in Uterovaginal Support." Am J Obstet Gynecol. 2020;222(5):427–436. doi:10.1016/j.ajog.2019.10.006
21. Donaldson K, Huntington A, De Vita R. "Mechanics of Uterosacral Ligaments: Current Knowledge, Existing Gaps, and Future Directions." Ann Biomed Eng. 2021;49(8):1788–1804. doi:10.1007/s10439-021-02755-6
22. Tan T, Cholewa NM, Case SW, De Vita R. "Micro-Structural and Biaxial Creep Properties of the Swine Uterosacral-Cardinal Ligament Complex." Ann Biomed Eng. 2016;44(11):3225–3237. doi:10.1007/s10439-016-1661-z
23. Renthal NE, Williams KC, Montalbano AP, et al. "Molecular Regulation of Parturition: A Myometrial Perspective." Cold Spring Harb Perspect Med. 2015;5(11):a023069. doi:10.1101/cshperspect.a023069
24. Mendelson CR, Gao L, Montalbano AP. "Multifactorial Regulation of Myometrial Contractility During Pregnancy and Parturition." Front Endocrinol. 2019;10:714. doi:10.3389/fendo.2019.00714
25. Renthal NE, Williams KC, Mendelson CR. "MicroRNAs — Mediators of Myometrial Contractility During Pregnancy and Labour." Nat Rev Endocrinol. 2013;9(7):391–401. doi:10.1038/nrendo.2013.96
26. Zangeneh FZ, Hantoushzadeh S. "The Physiological Basis With Uterine Myometrium Contractions From Electro-Mechanical/Hormonal Myofibril Function to the Term and Preterm Labor." Heliyon. 2023;9(11):e22259. doi:10.1016/j.heliyon.2023.e22259