The Bladder
The bladder is a distensible muscular reservoir that spends ~99% of its life storing urine and only 2–3 minutes per day emptying it.[1] For the reconstructive urologist this ratio is the whole point: the bladder's value is its low-pressure capacitance during storage and its coordinated, complete emptying when voluntary discharge is desired. Reconstructive decisions — continence procedures, outlet surgery, augmentation, diversion, ureteral reimplantation, neobladder — ultimately turn on preserving or rebuilding those two properties. This article focuses on the surgical anatomy, vascular and neural supply, and functional physiology that matter at the table, not a molecular account of urothelial sensory biology.
 OpenStax Anatomy & Physiology — The urinary bladder. (CC BY 4.0)
OpenStax Anatomy & Physiology — The urinary bladder. (CC BY 4.0)
Surgical Anatomy
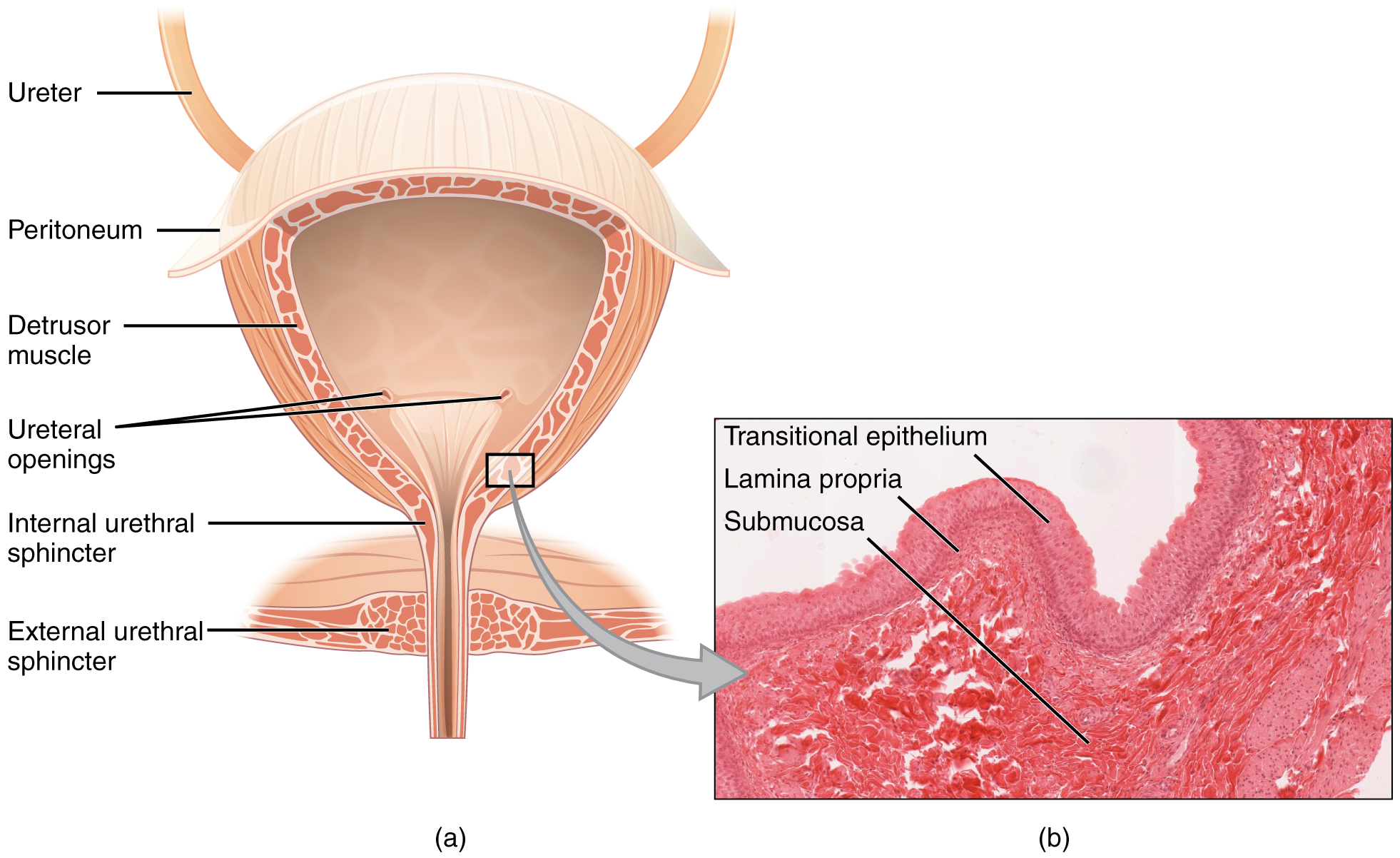
The adult bladder is a pelvic organ when empty, becoming intra-abdominal as it fills — a clinically relevant fact for suprapubic access, Pfannenstiel incisions, and penetrating trauma interpretation. Capacity is ~400–600 mL.[1]
Position and relations
| Aspect | Relation |
|---|---|
| Anterior | Pubic symphysis, retropubic space of Retzius (areolar plane exploited for suprapubic access, retropubic prostatectomy, Burch colposuspension, and many sling procedures) |
| Superior (dome) | Peritoneum, small bowel, sigmoid colon — the only peritonealized surface; intraperitoneal rupture occurs here |
| Posterior (M) | Vas deferens ampullae, seminal vesicles, rectum — rectovesical (Denonvilliers') fascia between |
| Posterior (F) | Anterior vaginal wall, uterine cervix (vesicouterine pouch extends to mid-bladder; anterior dissection in hysterectomy enters here) |
| Inferior | Prostate (M) or pelvic floor / urethra (F); bladder neck fixed to the pelvic floor |
| Lateral | Obturator internus and levator ani; neurovascular pedicles enter posterolaterally |
Gross structure
- Dome and body — thin-walled, most distensible region; the site where most diverticula and intraperitoneal ruptures occur.
- Trigone — triangular muscular floor between the two ureteral orifices and the internal urethral meatus. The trigone is less distensible, fixed to the pelvic floor, and is the structural substrate of the anti-reflux mechanism (see The Ureters).
- Bladder neck (vesical outlet) — where detrusor fibers converge and are continuous with smooth muscle of the proximal urethra. Forms the internal (involuntary) urethral sphincter.
Ligaments and the retropubic space
- Median umbilical ligament — obliterated urachus running from the dome to the umbilicus. Persistent urachal remnants produce cysts, sinuses, and adenocarcinoma.
- Medial umbilical ligaments — paired obliterated umbilical arteries; important landmarks entering the retropubic space laparoscopically or robotically.
- Puboprostatic (M) / pubovesical (F) ligaments — anchor the bladder neck to the pubis; preservation is part of most contemporary continence-sparing prostatectomy techniques.
- Lateral pedicles — vascular/neural pedicles attaching the bladder to the pelvic sidewall, carrying the inferior vesical vessels and bladder-branch pelvic plexus.
Vascular Supply and Drainage
Arterial supply
The bladder is supplied by branches of the internal iliac (hypogastric) artery:
| Vessel | Origin | Territory |
|---|---|---|
| Superior vesical artery | From the patent portion of the obliterated umbilical artery | Anterosuperior dome and lateral wall |
| Inferior vesical artery | Internal iliac anterior division (M; often gives prostatic branches) | Bladder base and trigone; seminal vesicles / prostate |
| Vaginal (F) / middle rectal / obturator (accessory) | Variable internal iliac branches | Bladder base in women; small accessory territories |
The standard cystectomy pedicle sequence — superior vesical then lateral then posterior (inferior vesical) — is built on this arterial geometry. The inferior vesical pedicle is the critical structure for both hemostasis and nerve sparing: the cavernosal / clitoral nerves travel immediately posterolateral to it as part of the inferior hypogastric (pelvic) plexus. Blind clamping of this pedicle is the commonest surgical cause of erectile or clitoral dysfunction after cystectomy.
Venous drainage
The vesical venous plexus drains to the internal iliac veins. In men it communicates freely with the prostatic (Santorini's) plexus anterior to the bladder neck and prostate — the engorged veins of this communication are the dominant bleeding source at dorsal vein / apical prostatic dissection and at cystectomy.
Lymphatic drainage
Lymphatic drainage follows the arteries to the obturator, external iliac, and internal iliac nodes, with secondary drainage to common iliac and presacral basins. Standard pelvic lymphadenectomy for muscle-invasive bladder cancer targets the obturator, external iliac, and internal iliac groups; extended templates add common iliac up to the aortic bifurcation and the presacral group.
Bladder Wall and Compliance
The wall has four layers, but for operative reasoning only two behaviors matter: the mucosal barrier and detrusor compliance.
| Layer | Composition | What matters operatively |
|---|---|---|
| Urothelium | Stratified transitional epithelium (3–6 cell layers; basal, intermediate, umbrella cells) | High-resistance barrier to water and solutes; also an active sensory surface that releases ATP, NO, and prostanoids in response to stretch — the neurophysiologic substrate of urgency sensation[3][4] |
| Lamina propria | ECM, elastic fibers, vessels, nerve endings, interstitial cells, muscularis mucosae | The plane of superficial (Ta/T1) tumor invasion; the communication hub between urothelium and detrusor[2] |
| Detrusor | Heterogeneously oriented smooth-muscle bundles functioning as a syncytium via gap junctions | The muscularis propria — invasion of this layer (≥T2) defines muscle-invasive bladder cancer and changes the operative plan entirely[5] |
| Adventitia / serosa | Fibrous coat laterally; peritoneum on the dome only | Intraperitoneal vs. extraperitoneal rupture determines management |
Compliance
Compliance = ΔV / ΔP — the change in volume per unit change in vesical pressure during filling. Normal values are >20 mL/cmH₂O; a compliance <10–12 mL/cmH₂O with filling pressures sustained above 40 cmH₂O is a recognized threshold for upper-tract risk (the "hostile bladder").
Compliance is reduced by:
- Chronic bladder outlet obstruction (detrusor hypertrophy and collagen deposition)
- Radiation cystitis and chronic inflammation (interstitial fibrosis)
- Chronic indwelling catheters
- Neurogenic dysfunction, particularly suprasacral spinal cord injury with upper motor neuron detrusor overactivity
Restoring compliance — by relieving obstruction, reducing overactivity, or surgically expanding the reservoir (augmentation cystoplasty, continent diversion) — is the core objective whenever the bladder is putting the kidneys at risk.
Innervation
The bladder is controlled by three peripheral neural systems acting in concert with the central switching circuits of the pontine micturition center (Barrington's nucleus).[6][7][10]
| System | Spinal origin | Nerve | Effect |
|---|---|---|---|
| Parasympathetic | S2–S4 | Pelvic | Detrusor contraction (M3 ≫ M2 muscarinic) — the voiding effector |
| Sympathetic | T10–L2 | Hypogastric (from superior hypogastric plexus) | Detrusor relaxation (β3-adrenergic, dome); bladder-neck contraction (α1-adrenergic, trigone and neck) — the storage effector |
| Somatic | S2–S4, Onuf's nucleus | Pudendal | External urethral sphincter contraction (voluntary) |
Clinical geometry of pelvic innervation
- The pelvic plexus (inferior hypogastric plexus) sits on the lateral rectum / posterolateral to the bladder base. Its bladder branches enter through the lateral vascular pedicles. Nerve-sparing in radical cystectomy and radical prostatectomy is anatomically possible because these fibers run immediately posterolateral to the inferior vesical vessels — ligation close to the bladder wall spares them; ligation wide on the pelvic sidewall does not.
- The pudendal nerve is sacrificed only in very extended pelvic procedures; in most cystectomy and prostatectomy work it is spared and external-sphincter function is preserved.
- Spinal cord lesion level predicts the bladder behavior pattern: suprapontine lesions (stroke, multiple sclerosis, Parkinson) disinhibit the PMC producing detrusor overactivity with preserved coordination; suprasacral spinal lesions (traumatic SCI above conus) produce detrusor–sphincter dyssynergia with high storage pressures; sacral / cauda equina / peripheral lesions produce an acontractile bladder with retention and overflow.
Referred pain
Visceral afferents from the bladder travel with the pelvic and hypogastric nerves to T10–L2 (sympathetic) and S2–S4 (parasympathetic) roots, with bladder-base and trigone discomfort referred to the suprapubic region and perineum. Bladder pain is poorly localized compared with cutaneous pain — a feature that makes interstitial cystitis and chronic pelvic pain syndromes notoriously hard to localize.
Storage and Voiding Physiology
Storage and voiding operate as two distinct reflex patterns switched at the pons.[1][6][10]
Storage (the default state)
- Sympathetic tone on — β3-mediated detrusor relaxation; α1-mediated bladder-neck tone.
- Somatic tone on — EUS tonically contracted via pudendal input from Onuf's nucleus.
- Parasympathetic tone off — no detrusor contraction.
- Guarding reflex — any transient rise in intravesical pressure (cough, sneeze, laugh, positional) triggers a spinal reflex that contracts the EUS more strongly — the mechanical basis of normal stress continence.[11]
Voiding (switched by the pons)
- Conscious decision and cortical permission release the tonic inhibition of the pontine micturition center.
- PMC output relaxes the EUS first, then drives parasympathetic pelvic-nerve output to contract the detrusor against a now-open outlet.
- Sympathetic storage tone is simultaneously inhibited.
- The micturition reflex operates in an all-or-none manner once triggered.[10][11]
When the switch fails
- Detrusor–sphincter dyssynergia (DSD) — loss of PMC coordination (SCI above the conus) produces simultaneous detrusor and sphincter contraction: high pressures, incomplete emptying, upper-tract risk. A surgical problem because medical management is usually insufficient to protect the kidneys long-term.
- Detrusor overactivity — involuntary detrusor contractions during filling; neurogenic (suprapontine/suprasacral) or idiopathic. The target of antimuscarinics, β3-agonists, intradetrusor onabotulinumtoxinA, and sacral neuromodulation.
- Underactive / acontractile detrusor — sacral-level or peripheral lesion; presents with retention and overflow. Managed with clean intermittent catheterization and, selectively, sacral neuromodulation or reconstruction.
- Bladder outlet obstruction — mechanical resistance (BPH, urethral stricture, bladder-neck contracture) raises voiding pressures and over time produces detrusor hypertrophy, trabeculation, diverticula, and reduced compliance. Surgical relief is definitive therapy.
Reference values
| Parameter | Typical adult value |
|---|---|
| Functional bladder capacity | 400–600 mL |
| First desire to void | ~150–200 mL |
| Normal compliance | >20 mL/cmH₂O |
| "Hostile" bladder threshold | Compliance <12 mL/cmH₂O with sustained storage pressures >40 cmH₂O |
| Post-void residual (normal) | <50–100 mL |
| Peak flow (Qmax), men | 15–25 mL/s |
| Peak flow (Qmax), women | 20–30 mL/s |
| Voided volume | ~300 mL per void; 6–8 voids per 24 h |
| Storage/voiding time ratio | ~99 : 1 |
Clinical Correlations for the Reconstructive Urologist
- Hostile bladder and upper-tract protection. Sustained detrusor leak-point pressure >40 cmH₂O predicts upper-tract deterioration independent of etiology; restoring low-pressure storage (pharmacologic or surgical, including augmentation cystoplasty and continent diversion) is the primary objective.
- Wall remodeling from obstruction. Chronic outlet obstruction causes detrusor hypertrophy, trabeculation, pseudodiverticula, and — eventually — true diverticula. True diverticula lack a muscular wall, empty poorly, and incubate stones, infection, and carcinoma. Diverticulectomy is indicated for recurrent UTI, stones, residual retention after outlet relief, or suspected neoplasm.
- Anti-reflux at the UVJ. The trigone is the fixed, less-distensible substrate against which the oblique submucosal ureter is compressed during filling. Trigonal resection (extensive T2 tumors, some augmentations) removes this platform; reimplantation or neobladder design must recreate it. See The Ureters for the 5 : 1 tunnel-to-diameter rule.
- Nerve-sparing in cystectomy and prostatectomy. The pelvic plexus runs immediately posterolateral to the inferior vesical / prostatic pedicles. Ligation close to the bladder and prostate spares cavernosal / clitoral input and preserves sexual function; wider sidewall ligation does not.
- Lymphadenectomy templates. Obturator, external iliac, and internal iliac basins drain the bladder primarily; standard LND in muscle-invasive cancer covers these, with extended templates reaching the common iliac and presacral regions.
- Neurogenic bladder by lesion level. Suprapontine lesions produce OAB with coordinated voiding; suprasacral cord lesions produce DSD and high-pressure storage — the highest upper-tract-risk phenotype; sacral / cauda equina / peripheral lesions produce acontractile bladder with retention. Management algorithms follow these three patterns and branch on storage pressure.
- Bladder-neck competence matters after prostatectomy. The internal (involuntary) urethral sphincter is the bladder neck. In most radical prostatectomies it is incorporated into the urethrovesical anastomosis; continence after surgery therefore depends on the external sphincter and on anatomical preservation of the striated-sphincter complex and its innervation.
- Urachal / umbilical abnormalities. Urachal remnants (cysts, sinuses, patent urachus) present across the lifespan; urachal adenocarcinoma arises from the dome / anterior wall and typically requires partial cystectomy en bloc with the urachus and umbilicus.
- Rupture and trauma. Intraperitoneal rupture (typically dome, after blunt trauma to a full bladder) requires operative repair; extraperitoneal rupture (typically from pelvic-fracture shears at the bladder neck) is usually managed with catheter drainage unless associated with bone fragments in the bladder, bladder-neck involvement, or concurrent abdominal surgery.
- Sensory signaling in IC/BPS and OAB. The urothelium is a sensor as well as a barrier; altered release of ATP, prostanoids, and NO and altered afferent responsiveness underlie urgency in OAB and the pain of interstitial cystitis / bladder pain syndrome. Intradetrusor onabotulinumtoxinA likely acts partly on these afferent pathways in addition to the motor effect.[4][8][9]
Videos
References
1. Panicker JN, Fowler CJ, Kessler TM. "Lower Urinary Tract Dysfunction in the Neurological Patient: Clinical Assessment and Management." Lancet Neurol. 2015;14(7):720–732. doi:10.1016/S1474-4422(15)00070-8
2. Andersson KE, McCloskey KD. "Lamina Propria: The Functional Center of the Bladder?" Neurourol Urodyn. 2014;33(1):9–16. doi:10.1002/nau.22465
3. Dalghi MG, Montalbetti N, Carattino MD, Apodaca G. "The Urothelium: Life in a Liquid Environment." Physiol Rev. 2020;100(4):1621–1705. doi:10.1152/physrev.00041.2019
4. Birder L, Andersson KE. "Urothelial Signaling." Physiol Rev. 2013;93(2):653–680. doi:10.1152/physrev.00030.2012
5. Fry CH, McCloskey KD. "Spontaneous Activity and the Urinary Bladder." Adv Exp Med Biol. 2019;1124:121–147. doi:10.1007/978-981-13-5895-1_5
6. de Groat WC. "Integrative Control of the Lower Urinary Tract: Preclinical Perspective." Br J Pharmacol. 2006;147 Suppl 2:S25–S40. doi:10.1038/sj.bjp.0706604
7. Wecht JM, Krassioukov AV, Alexander M, et al. "International Standards to Document Autonomic Function Following SCI (ISAFSCI): Second Edition." Top Spinal Cord Inj Rehabil. 2021;27(2):23–49. doi:10.46292/sci2702-23
8. Merrill L, Gonzalez EJ, Girard BM, Vizzard MA. "Receptors, Channels, and Signalling in the Urothelial Sensory System in the Bladder." Nat Rev Urol. 2016;13(4):193–204. doi:10.1038/nrurol.2016.13
9. Heppner TJ, Fallon HC, Rengo JL, et al. "Urothelium-Derived Prostanoids Enhance Contractility of Urinary Bladder Smooth Muscle and Stimulate Bladder Afferent Nerve Activity in the Mouse." Am J Physiol Regul Integr Comp Physiol. 2024;327(1):R97–R108. doi:10.1152/ajpregu.00084.2024
10. de Groat WC, Griffiths D, Yoshimura N. "Neural Control of the Lower Urinary Tract." Compr Physiol. 2015;5(1):327–396. doi:10.1002/cphy.c130056
11. Danziger ZC, Grill WM. "Sensory Feedback From the Urethra Evokes State-Dependent Lower Urinary Tract Reflexes in Rat." J Physiol. 2017;595(16):5687–5698. doi:10.1113/JP274191