Male Urethra
The male urethra is a ~18–20 cm muscular conduit that carries urine from the bladder neck to the external meatus and, during ejaculation, semen to the outside — two functions that impose competing demands on its sphincteric, neural, and mucosal biology.[1] For the reconstructive urologist it is the central operative field: urethral stricture disease, pelvic-fracture urethral injury (PFUI), post-prostatectomy incontinence, and hypospadias/epispadias reconstruction all turn on a precise understanding of its segmental anatomy, dual sphincter complex, and vascular and neural supply. This article focuses on those details, not on molecular sensory biology.
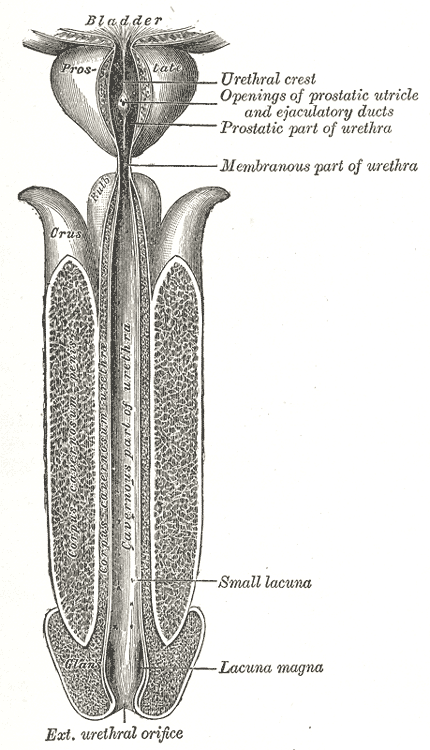 Gray's Anatomy Fig. 1142 — The male urethra laid open on its anterior (upper) surface, showing the prostatic, membranous, and penile segments.
Gray's Anatomy Fig. 1142 — The male urethra laid open on its anterior (upper) surface, showing the prostatic, membranous, and penile segments.
Segmental Anatomy
The urethra divides both anatomically (by the structure it traverses) and functionally (into posterior and anterior segments); both divisions matter for operative planning.[1][2]
| Segment | Length | Course | Surgical relevance |
|---|---|---|---|
| Prostatic | 3–4 cm | Through the prostate from bladder neck to verumontanum; receives the ejaculatory ducts | Disrupted by TURP/HoLEP/simple prostatectomy; the site of post-TURP bladder-neck contracture; proximal reconstruction target in PFUI |
| Membranous | 1–2 cm | Through the perineal membrane / urogenital diaphragm; narrowest point of the urethra | Surrounded by the rhabdosphincter — the dominant continence structure after prostatectomy; shearing point in pelvic-fracture urethral injury |
| Bulbar | ~5–6 cm | Within the bulb of the corpus spongiosum in the perineum | The most common stricture location; main working segment of anastomotic and augmentation urethroplasty |
| Penile (pendulous) | ~10 cm | Through the penile shaft within the corpus spongiosum | Stricture site after instrumentation/hypospadias repair; substitution urethroplasty territory |
| Fossa navicularis | Terminal | Dilated segment within the glans | Meatal stenosis and distal stricture site; lichen-sclerosus territory |
Functionally the urethra is divided at the urogenital diaphragm into:
- Posterior urethra — prostatic + membranous; posterior, supported by the perineal membrane and the rhabdosphincter; drains to pelvic nodes.
- Anterior urethra — bulbar + penile + fossa navicularis; contained within the corpus spongiosum; drains to inguinal nodes.
This anterior/posterior split governs lymphatic drainage for urethral cancer staging, trauma mechanism (straddle vs pelvic-fracture shears), and reconstruction approach (perineal vs retropubic/trans-pubic access for PFUI).
Mucosal Biology
The epithelial lining transitions along the length:[1][4][5][7]
| Segment | Epithelium |
|---|---|
| Prostatic (proximal) | Urothelium, then a small zone of prostatic glandular epithelium |
| Membranous and bulbar | Pseudostratified columnar |
| Penile | Pseudostratified columnar transitioning distally |
| Fossa navicularis and meatus | Non-keratinized stratified squamous, glycogen-rich, estrogen-responsive |
Two operative consequences:
- The fossa navicularis is squamous and estrogen-responsive[5] — which is why it is the preferred territory for lichen sclerosus and why topical estrogen has an adjunctive role in certain distal stricture regimens.
- Minor prostatic glands scattered along the urethra[4] explain the occasional finding of prostatic-type tissue in ectopic locations on reconstructive histology.
The urethra is glandular throughout:
- Glands of Littré — mucus-secreting glands along the penile and bulbar urethra. Their inflammation is the structural starting point of most idiopathic bulbar strictures (see below).
- Cowper's (bulbourethral) glands — lie in the urogenital diaphragm lateral to the membranous urethra; ducts empty into the proximal bulbar urethra.
- Lacuna magna — a dorsal pocket in the fossa navicularis; site of occasional irritative symptoms in children.
Sphincter Complex
Male continence is maintained by two sphincters with complementary temporal profiles:[4][9][10][13]
| Sphincter | Muscle type | Location | Role |
|---|---|---|---|
| Lissosphincter (internal, smooth) | Autonomic smooth muscle; myogenic tone via L-type Ca²⁺ channels | Bladder neck → prostatic urethra | Tonic, passive continence at rest; closes during ejaculation to prevent retrograde flow |
| Rhabdosphincter (external, striated) | Voluntary striated muscle (Onuf's nucleus, S2–S4 via pudendal nerve) | Membranous urethra, extending over the prostate apex as an omega-shaped loop anterolaterally | Phasic, active continence during cough/Valsalva; the dominant continence structure after radical prostatectomy |
Two key anatomic facts drive operative decisions:
- The rhabdosphincter is not circumferential; it is an anterolateral omega wrapping around the membranous urethra with a deficient posterior portion supplied by the levators and posterior median fibrous raphe.[4][9] Lateral dissection at the apex therefore threatens its bulk and its innervation.
- The rhabdosphincter contains nearly equal slow- and fast-twitch fibers, which is why it sustains tonic closure pressure and can respond phasically.
Continence after radical prostatectomy depends on preserved membranous urethral length, intact rhabdosphincter innervation, and preserved pelvic support (pubourethral ligaments, puboperinealis). All three are potentially modifiable at the time of surgery — long membranous urethra on preoperative MRI, apical nerve sparing, and preservation of the puboprostatic ligaments are each independently associated with earlier return of continence.
Innervation
The male urethra receives autonomic, somatic, and sensory input, arriving along two anatomically distinct routes that are both clinically exploited and clinically violated.[8][11][12][13]
Somatic
- Pudendal nerve (S2–S4), via Onuf's nucleus, supplies the rhabdosphincter and levator ani. Approximately 70% of external-sphincter closure pressure derives from S3; S2 and S4 contribute the remainder.[12]
- Clinical corollary: unilateral sacral root injury preserves continence in most patients; bilateral S3 injury (cauda equina, complex sacral resection) is the typical cause of surgically induced sphincteric incontinence.
Autonomic
- Sympathetic (T10–L2) via the hypogastric nerve and pelvic plexus — α₁-adrenergic tone closes the bladder neck and proximal urethra during storage and ejaculation; β-adrenergic fibers contribute relaxation distally.[2][10]
- Parasympathetic (S2–S4) via the pelvic nerve — modulates urethral smooth muscle with cholinergic and nitrergic fibers; nitric oxide is the dominant inhibitory transmitter and is responsible for urethral relaxation at the onset of voiding.
Dual origin of urethral and cavernosal nerves
The nerves to the urethra and corpora cavernosa arise from the pelvic plexus and reach the urethra through two anatomically distinct pathways:[7]
- Dorsal route — along the ejaculatory ducts and seminal vesicles to the prostate base and proximal urethra.
- Lateral route — along the inferior vesical artery, forming the neurovascular bundle (NVB) at the posterolateral aspect of the prostate.
Both routes must be respected during nerve-sparing prostatectomy, pelvic-fracture urethral repair, and rectourethral fistula repair — preserving only the lateral NVB does not preserve all cavernosal input.
Sensory
Sensory afferents travel with the pudendal (somatic) and pelvic (visceral) nerves. Mechano- and chemosensitive feedback from the urethra contributes to state-dependent lower-urinary-tract reflexes — urethral flow at low bladder volume reinforces sphincteric guarding; at high volume it augments voiding.[14][15]
Vascular Supply
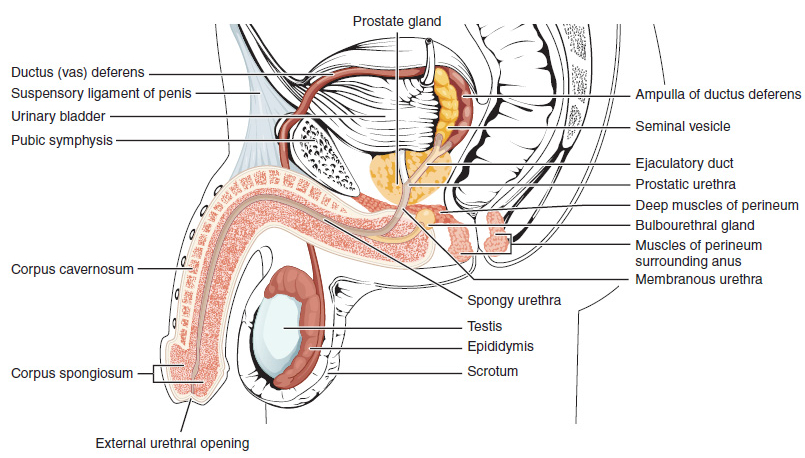 Lateral cross-section of the penis demonstrating the urethra within the corpus spongiosum, flanked by the paired corpora cavernosa.
Lateral cross-section of the penis demonstrating the urethra within the corpus spongiosum, flanked by the paired corpora cavernosa.
Urethral blood supply is segmental and bidirectional, with clinically important redundancy. This redundancy is the reason the urethra can be mobilized for reconstruction — and the reason each reconstructive technique specifies which feeders must be preserved.
| Segment | Dominant supply | Clinical note |
|---|---|---|
| Prostatic / bladder neck | Inferior vesical artery, prostatic branches | Disrupted by bladder-neck contracture after TURP or radiation |
| Membranous | Perineal and inferior vesical contributions | Watershed at the urogenital diaphragm |
| Bulbar | Bulbar artery (branch of internal pudendal) + retrograde flow from dorsal and urethral arteries | The retrograde flow is what allows non-transecting anastomotic urethroplasty to survive on distal dorsal-artery input; transecting operations depend on intact retrograde supply |
| Penile | Urethral artery (branch of internal pudendal) — paired longitudinal vessels within the corpus spongiosum | Cavernosal dissection in penile urethroplasty must preserve these |
| Glans / fossa navicularis | Dorsal artery anastomosis | Preserved by dorsal onlay and dorsal-inlay techniques |
Venous drainage parallels the arteries; the deep dorsal vein of the penis drains the penile and glandular urethra into the prostatic (Santorini's) plexus and thence the internal iliac system.
Physiology — Storage, Voiding, and Ejaculation
Storage
- Tonic lissosphincter tone holds the bladder neck closed.
- Rhabdosphincter tone (fatigue-resistant slow-twitch fibers) maintains a maximal urethral closure pressure >100 cmH₂O in young healthy men, declining with age.
- On cough or Valsalva, reflex recruitment of fast-twitch rhabdosphincter fibers increases closure pressure faster than intra-abdominal pressure rises — the functional basis of stress continence.
Voiding
Recent high-resolution imaging data suggest the urethra does not passively open with detrusor contraction; rather, the anterior and lateral urethral muscle unit actively shortens and opens the lumen from the start of micturition.[6] In practical terms:
- Somatic (pudendal) tone drops first.
- Parasympathetic nitrergic output relaxes urethral smooth muscle and opens the lumen.
- Sympathetic α-tone is simultaneously withdrawn.
- Only then does detrusor contraction empty the bladder against a now-open outlet.
Failure of this sequence produces detrusor–sphincter dyssynergia (see The Bladder).
Ejaculation
- Sympathetic α-tone closes the bladder neck to prevent retrograde ejaculation — the mechanism disrupted by TURP, α-blockers (tamsulosin, silodosin), and bladder-neck-incising procedures.
- Rhabdosphincter relaxes briefly for emission, then undergoes rhythmic contraction to propel semen antegrade.
Clinical Correlations for the Reconstructive Urologist
- Stricture geography. The bulbar urethra is the commonest stricture location; inflammation in the glands of Littré drives spongiofibrosis in the surrounding corpus spongiosum, and the depth of spongiofibrosis — not the length of the stricture alone — determines whether non-transecting repair, excision-and-primary-anastomosis (EPA), or substitution urethroplasty (buccal-graft onlay / inlay) is appropriate.
- Pelvic-fracture urethral injury (PFUI). PFUI complicates 1.5–5% of anterior pelvic fractures; risk increases by approximately 10% for every 1 mm of pubic symphysis diastasis.[3][8b] The injury is a shear at the membranous urethra against the fixed prostatic apex, producing a distraction defect. Delayed anastomotic urethroplasty (after ~3 months) is the gold standard; acute realignment is an adjunct, not a definitive repair.
- Post-prostatectomy incontinence. Mediated by shortened membranous urethra, rhabdosphincter or pudendal-branch injury, loss of puboprostatic support, or a concomitant hyperactive bladder. AUS implantation is the gold standard for moderate-to-severe persistent SUI; male slings are appropriate for mild-to-moderate SUI in non-irradiated patients with preserved detrusor contractility.
- Post-TUR bladder-neck contracture. Arises from disruption of the lissosphincter and proximal urethral mucosa at TURP; presents as a ring at the bladder neck and is managed with endoscopic incision, sometimes with adjunctive steroid injection or, in refractory cases, open reconstruction.
- Lymphatic drainage determines cancer workup. Anterior urethral cancers drain to superficial and deep inguinal nodes; posterior urethral cancers drain to pelvic (external iliac, obturator) nodes. Inguinal nodal assessment and the staging approach change accordingly.
- Retrograde ejaculation. Iatrogenic bladder-neck incompetence (α-blockers, TURP, bladder-neck incision, retropubic prostatectomy) abolishes α₁-mediated neck closure; counseling for fertility should precede these procedures.
- Nerve-sparing in PFUI and rectourethral fistula repair. The dual (dorsal + lateral/NVB) route of cavernosal innervation means that neither pathway alone preserves full erectile function; technique must protect both where the injury allows.
- Lichen sclerosus and the squamous distal urethra. LS preferentially involves the glans, meatus, and fossa navicularis — the squamous, estrogen-responsive territory. Progressive involvement drives meatal stenosis and retrograde scarring; definitive reconstruction favors non-genital skin or buccal-graft substitution rather than local skin flaps in affected territory.
Videos
References
1. Carroll PR, Dixon CM. "Surgical Anatomy of the Male and Female Urethra." Urol Clin North Am. 1992;19(2):339–346.
2. Martin JM, Richardson M, Siva S, et al. "Mechanisms, Mitigation, and Management of Urinary Toxicity From Prostate Radiotherapy." Lancet Oncol. 2022;23(12):e534–e543. doi:10.1016/S1470-2045(22)00544-7
3. Johnsen N, Wessells H, Archer-Arroyo K, et al. "Best Practices Guidelines — Management of Genitourinary Injuries." American College of Surgeons. 2025.
4. Cohen RJ, Garrett K, Golding JL, Thomas RB, McNeal JE. "Epithelial Differentiation of the Lower Urinary Tract With Recognition of the Minor Prostatic Glands." Hum Pathol. 2002;33(9):905–909. doi:10.1053/hupa.2002.127440
5. Holstein AF, Davidoff MS, Breucker H, Countouris N, Orlandini G. "Different Epithelia in the Distal Human Male Urethra." Cell Tissue Res. 1991;264(1):23–32. doi:10.1007/BF00305719
6. Watanabe H, Takahashi S, Ukimura O. "Urethra Actively Opens From the Very Beginning of Micturition: A New Concept of Urethral Function." Int J Urol. 2014;21(2):208–211.
8b. Coccolini F, Moore EE, Kluger Y, et al. "Kidney and Uro-Trauma: WSES-AAST Guidelines." World J Emerg Surg. 2019;14:54. doi:10.1186/s13017-019-0274-x
7. Röthlisberger R, Aurore V, Boemke S, et al. "The Anatomy of the Male Inferior Hypogastric Plexus: What Should We Know for Nerve Sparing Surgery." Clin Anat. 2018;31(6):788–796.
8. Akita K, Sakamoto H, Sato T. "Origins and Courses of the Nervous Branches to the Male Urethral Sphincter." Surg Radiol Anat. 2003;25(5–6):387–392.
9. Koraitim MM. "The Male Urethral Sphincter Complex Revisited: An Anatomical Concept and Its Physiological Correlate." J Urol. 2008;179(5):1683–1689.
10. Sergeant GP, Hollywood MA, Thornbury KD. "Spontaneous Activity in Urethral Smooth Muscle." Adv Exp Med Biol. 2019;1124:149–167. doi:10.1007/978-981-13-5895-1_6
11. Wecht JM, Krassioukov AV, Alexander M, et al. "International Standards to Document Autonomic Function Following SCI (ISAFSCI): Second Edition." Top Spinal Cord Inj Rehabil. 2021;27(2):23–49. doi:10.46292/sci2702-23
12. Juenemann KP, Lue TF, Schmidt RA, Tanagho EA. "Clinical Significance of Sacral and Pudendal Nerve Anatomy." J Urol. 1988;139(1):74–80.
13. Yashiro K, Thor KB, Burgard EC. "Properties of Urethral Rhabdosphincter Motoneurons and Their Regulation by Noradrenaline." J Physiol. 2010;588(Pt 24):4951–4967.
14. Danziger ZC, Grill WM. "Sensory Feedback From the Urethra Evokes State-Dependent Lower Urinary Tract Reflexes in Rat." J Physiol. 2017;595(16):5687–5698. doi:10.1113/JP274191
15. Hinata N, Murakami G, Abe S, et al. "Detailed Histological Investigation of the Female Urethra: Application to Radical Cystectomy." J Urol. 2012;187(2):451–456. doi:10.1016/j.juro.2011.10.037