Cesarean Section
Cesarean delivery is one of the most commonly performed operations in the world — 32.2% of U.S. births in 2022 were cesarean,[1] and global rates continue to rise. The reconstructive urologist and urogynecologist are not the surgeons performing this operation, but we are the surgeons who repair the injuries it produces. A meaningful share of acquired bladder injuries, vesicouterine fistulas (Youssef syndrome), ureteral injuries, and uterine isthmocele-related symptomatology in women of reproductive age originate at cesarean delivery. Understanding the operation — specifically where the dissection travels, which layer holds where, and which steps place the urinary tract at risk — is prerequisite to repairing its complications well.
This article covers cesarean delivery from the reconstructive surgeon's perspective: indications, the major technical variants and their evidence base, the anatomic danger zones for urinary-tract injury, the spectrum of GU injuries that follow cesarean (including the particular geometry of post-cesarean vesicouterine fistula), and how the original operation shapes the repair strategy we choose.
See also: The Bladder, The Uterus, The Cervix, Pelvic Vascular Anatomy, Incisions & Closure.
Epidemiology
Cesarean delivery rates have risen from <5% in 1970 to approximately one-third of U.S. births today, with substantial regional variation (Latin America >40%; Sub-Saharan Africa <5%).[1] The consequence for reconstructive practice is cumulative: every additional cesarean increases adhesion burden, thins the lower uterine segment, fuses the bladder to the uterine scar, and raises the risk of placenta accreta spectrum in subsequent pregnancies — the single disorder in obstetrics most likely to result in massive lower-urinary-tract injury.[18][19]
Urinary tract injury at cesarean is uncommon in absolute terms but meaningful across the denominator:
| Injury | Rate at primary CD | Rate at repeat CD | Rate in CD for PAS |
|---|---|---|---|
| Bladder injury (any) | 0.08–0.3% | 0.6–1.0% | 12–29% |
| Ureteral injury | 0.03–0.09% | 0.1–0.3% | 2–7% |
| Vesicouterine fistula | Rare | Low | Higher after repeat CD |
Adhesion burden compounds with each operation. Observational series report pelvic adhesions in 12–46% after a second cesarean and 26–75% after a third, with secondary risk of small bowel obstruction and technical difficulty at any subsequent pelvic surgery (including GU reconstruction).[18]
Indications
A working familiarity with indications matters because it defines the operative context — an emergency cesarean for fetal distress through an unlabored lower uterine segment is a different operation than a scheduled repeat for placenta accreta. Primary-cesarean indications distribute approximately as follows:[2][3]
- Labor dystocia — 35–41% (most common)
- Abnormal or indeterminate fetal heart rate tracing — 23%
- Fetal malpresentation (breech, transverse lie) — ~10%
- Multiple gestation — a small but growing share
- Suspected fetal macrosomia — small share
- Absolute indications: placenta previa, placenta accreta spectrum, complete cord prolapse, active genital HSV, HIV with high viral load
Repeat cesarean now drives a substantial fraction of all cesarean volume, because a prior cesarean remains the strongest single predictor of a subsequent cesarean.
Surgical Technique
Abdominal Incision
Two transverse lower-abdominal approaches dominate. The Pfannenstiel incision — introduced 1900 — is curvilinear, 2 cm above the symphysis, with sharp dissection of subcutaneous tissue, transverse incision of the rectus fascia, and separation of the rectus muscles off the fascia by sharp and blunt technique. The Joel-Cohen incision — popularized for cesarean in the 1990s — is a straight transverse skin incision 3 cm below the line joining the anterior superior iliac spines, followed by blunt dissection of subcutaneous fat, a small central fascial cut extended laterally by finger traction, and blunt finger-stretching of the rectus muscles and peritoneum.[4][5][6]
The Joel-Cohen technique is favored by the bulk of current evidence. A Cochrane review found Joel-Cohen (vs Pfannenstiel) delivered:[4]
- 65% reduction in postoperative febrile morbidity (RR 0.35, 95% CI 0.14–0.87)
- Reduced postoperative pain and analgesic requirement (RR 0.55, 95% CI 0.40–0.76)
- Less blood loss (MD −58 mL)
- Shorter operating time (MD −11.4 minutes)
- Shorter skin-to-delivery time (MD −1.9 minutes)
- Shorter hospital stay (MD −1.5 days)
Relevance to reconstruction: the Pfannenstiel's more extensive subfascial dissection creates the dense suprapubic/prevesical adhesion plane that reconstructive urologists routinely encounter when approaching the bladder for fistula repair or bladder-neck reconstruction after prior cesarean. The higher, straighter Joel-Cohen incision produces less suprapubic scarring but the same intraperitoneal adhesion pattern. A vertical midline cesarean — now uncommon outside of massive obstetric hemorrhage, maternal cardiac arrest, or gravid uterus above the umbilicus — is associated with more significant incisional hernia and a different reoperation landscape.
Entry into the Peritoneal Cavity — First Point of Bladder Risk
Opening the parietal peritoneum is the first moment at which the bladder is at risk. In a term gravida with a distended lower uterine segment and a dome of bladder that may sit unusually high, sharp peritoneal entry without tenting can directly lacerate the bladder dome — especially in a patient with prior cesarean where the bladder is adherent to the abdominal wall or to the prior uterine scar. The recommended maneuver is to open the peritoneum at its most cephalad extent (closest to the umbilicus), tent it with two forceps, and incise transversely between them, then extend inferiorly under direct vision.
Reconstructive lesson: approximately 20–50% of all cesarean bladder injuries occur at peritoneal entry in the setting of prior cesarean, where the bladder is densely adherent to the scarred anterior uterine wall.[20][21] A high, tented entry is the single most effective prevention.
The Bladder Flap
The vesicouterine peritoneal reflection is incised and the bladder is mobilized inferiorly off the lower uterine segment to expose the hysterotomy site — the classic "bladder flap." The flap is developed in the loose areolar plane between bladder wall and uterine serosa.
A longstanding debate concerns whether the bladder flap should be routinely created at all. Randomized and meta-analytic evidence suggests omitting the bladder flap shortens operative and skin-to-delivery time, reduces blood loss, and does not increase bladder injury in low-risk primary cesarean with an undeveloped lower uterine segment.[5][6] In repeat cesarean and cesarean for labor at full dilation, however, the flap is often essential to protect the bladder — precisely because the bladder rides high on a scarred lower segment.
Mechanism of injury at the bladder flap: dissection outside the correct plane (too anterior — into the bladder wall itself; too posterior — into the myometrium with bleeding that obscures further dissection). Prior cesarean obliterates the plane entirely; sharp dissection in this setting, without clear entry into the areolar space, is the dominant mechanism of intraoperative cystotomy during repeat cesarean.
Hysterotomy
The standard hysterotomy is low transverse, made through the lower uterine segment 1–2 cm above the superior margin of the bladder. A small central incision is made with the scalpel down to — but not through — the membranes; the incision is then extended laterally by blunt finger traction (Joel-Cohen era) rather than by sharp extension with bandage scissors, which is associated with more cephalad-caudad angular tears into the lateral uterine vessels and broad ligament. Blunt extension along the natural muscle-fiber orientation reduces blood loss and extension-related injury.[5][6]
Alternative hysterotomies:
- Low vertical — a short vertical incision in the lower uterine segment. Used when the lower segment is narrow (breech presentation, transverse lie, very preterm) or when extension is anticipated. Can extend into the body (classical) if control is lost.
- Classical — midline vertical through the uterine body. Used for severe adhesions obliterating the lower segment, back-down transverse lie, some preterm deliveries, and most placenta previa with accreta. Carries a 4–9% risk of uterine rupture in subsequent pregnancy (vs <1% for low transverse), and mandates cesarean for all future deliveries.
Reconstructive relevance: the lower-transverse hysterotomy is the incision that later forms the isthmocele / niche — a wedge-shaped defect in the anterior lower uterine segment at the prior scar — and that defines the geometry of post-cesarean vesicouterine fistula. The low-transverse scar sits directly behind the trigone and interureteric ridge; a fistula at this location presents clinically as cyclic menouria (Youssef syndrome) because the trigone remains above the fistula and continent urinary storage is preserved.
Delivery of the Fetus and Placenta
The presenting part is delivered by traction and fundal pressure (for cephalic) or by internal podalic version and breech extraction (for transverse lie). The placenta is delivered either by spontaneous expulsion with gentle cord traction (preferred — reduced endometritis, shorter operative time) or by manual extraction (associated with higher endometritis and greater blood loss). Placenta accreta spectrum (PAS) is suspected when the placenta does not spontaneously separate and refuses to cleave; attempting forcible manual extraction in true accreta is the single most dangerous intraoperative step in obstetrics and the dominant mechanism of massive bladder injury, ureteral injury, and cesarean hysterectomy. See the PAS section below.
Uterine Closure
The question of single-layer vs double-layer closure has undergone substantial evidence turnover and remains an area of ongoing nuance. Traditional teaching favored double-layer closure on the theory that a thicker scar better resists subsequent uterine rupture. Contemporary evidence complicates that framing:
- A 2025 meta-analysis of 11 RCTs (6,058 participants) found single-layer closure associated with significantly lower niche prevalence at 3 months (RD −0.02) and 6 months (RD −0.11) compared to double-layer closure.[7]
- The 2Close trial (n = 2,292) found no superiority of double-layer closure for postmenstrual spotting; operative time was 3.9 minutes longer and niche prevalence 4.7% higher with double-layer.[8]
- At 3-year follow-up, the 2Close cohort showed no differences in live birth rates, pregnancy rates, or uterine rupture between techniques.[9]
- The CORONIS trial (n = 15,935) found no clear short-term benefits of double- over single-layer closure.[10]
A contemporary refinement, most fully articulated by Bujold & Romero (2026), argues that the closure's anatomic fidelity matters more than the number of layers: the endometrium should be excluded from the first layer to prevent endometrial-tissue entrapment within the myometrial scar (a presumed driver of niche formation and adenomyosis-like scar pathology), and the overlying myometrium should be reapproximated without ischemic bites.[11] An endometrium-free closure technique has been described and studied in the same AJOG cesarean supplement.[23]
Reconstructive relevance: the uterine niche (isthmocele) is a wedge-shaped defect in the anterior lower uterine segment at the prior hysterotomy scar. It presents with postmenstrual spotting, secondary infertility, cyclic pelvic pain, and — in a pregnant patient — as the substrate for a cesarean scar ectopic pregnancy or cesarean scar defect–associated placenta accreta. Reconstructive approach to the niche (hysteroscopic resection, laparoscopic niche repair, vaginal repair) is an evolving URPS subspecialty domain.
Peritoneal and Fascial Closure
- Visceral and parietal peritoneum: non-closure is now standard. Peritoneal closure does not reduce adhesions or morbidity and prolongs operative time.[10][12]
- Rectus muscle reapproximation: not routinely required.
- Fascial closure: running non-locking delayed-absorbable suture (e.g., 0 or 1 looped PDS / Maxon) in a Pfannenstiel; small-bites technique reduces incisional hernia rates.
- Subcutaneous closure: closure of Scarpa fascia reduces wound seroma and the "cesarean apron" (abdominal panniculus deformity) without increasing SSI.[24]
- Skin: subcuticular suture reduces wound-complication rates compared with staples.
Uterine Exteriorization
Delivery of the uterus through the incision for repair versus in-situ closure has no clear outcome difference. Exteriorization may slightly reduce blood loss and operative time at the cost of more intraoperative nausea and vomiting; in-situ repair avoids the hemodynamic perturbation but makes hemostasis at the angles more technically demanding.[12][13]
Anatomic Danger Zones
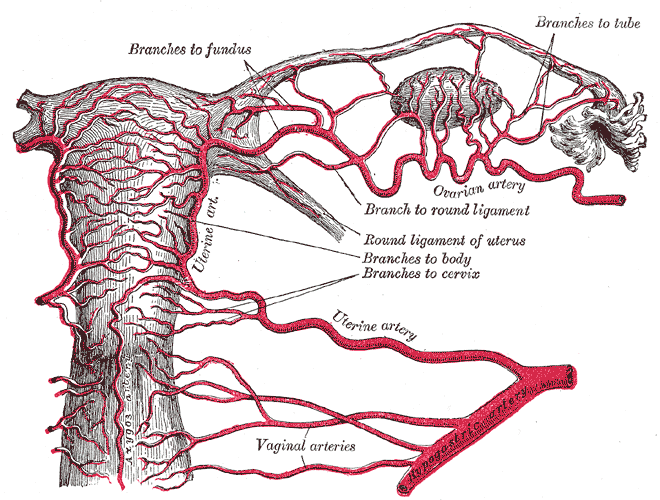 Uterus and broad ligament — the parametrial space houses the uterine artery, ureter, and branches of the internal iliac system. Gray's Anatomy of the Human Body (1918), public domain.
Uterus and broad ligament — the parametrial space houses the uterine artery, ureter, and branches of the internal iliac system. Gray's Anatomy of the Human Body (1918), public domain.
Palacios-Jaraquemada et al. (AJOG, 2026) provide the most current surgical-anatomy reference specifically aimed at routine and complex cesarean delivery; their framing identifies five anatomic domains that every cesarean surgeon must know.[25] For the reconstructive surgeon, the parametrial space, vesicouterine pouch, and retropubic space are the three domains where the urinary tract is most at risk.
The Vesicouterine Pouch
The bladder dome and anterior bladder wall sit immediately anterior to the lower uterine segment, separated by the vesicouterine peritoneal reflection and the loose areolar tissue of the vesicouterine space. In the unoperated patient this plane is bloodlessly developed by blunt dissection. After prior cesarean, the plane is obliterated by scar — the bladder dome may sit 3–5 cm higher than expected, fused to the anterior lower uterine segment. In placenta accreta spectrum with anterior placentation, the bladder wall itself may be invaded by placental tissue.
The Parametrial Space
The parametrium is the connective-tissue compartment lateral to the cervix and lower uterine segment. It houses:
- The uterine artery (arising from the anterior division of the internal iliac) crossing over the ureter ("water under the bridge") approximately 1.5–2 cm lateral to the cervix.
- The ureter, traveling inferomedially toward the trigone within the cardinal ligament.
- Branches of the internal iliac system (uterine, vaginal, cervicovaginal arteries).
- Dense lymphatics and autonomic nerve plexuses (inferior hypogastric plexus).
Lateral extension of the hysterotomy — an uncontrolled tear during blunt extension of the uterine incision — is the dominant mechanism of cesarean uterine artery injury and, less commonly, cesarean ureteral injury. The injury occurs where the tear runs lateral into the broad ligament, placing a hemorrhaging vessel in direct anatomic continuity with the ureter.
The Retropubic Space
The retropubic (prevesical) space of Retzius sits between the pubic symphysis anteriorly and the bladder posteriorly. Dissection here is relevant when the bladder must be mobilized off the symphysis — rare at primary cesarean, more common in repeat cesarean with a densely scarred suprapubic plane, and routine in cesarean hysterectomy for PAS. The retropubic space is the same space the reconstructive surgeon enters for Burch urethropexy, retropubic sling placement, and retropubic bladder-neck reconstruction — and prior cesarean obliteration of this space is a documented source of technical difficulty in those operations.
The Ureter at Cesarean
The ureter is rarely at risk during a standard low-transverse cesarean with a well-developed lower segment. Injury, when it occurs, is concentrated in the following settings:
- Lateral hysterotomy extension into the broad ligament, with uterine-artery injury and blind suture placement for hemostasis catching the ureter.
- Cesarean hysterectomy, particularly emergency hysterectomy for postpartum hemorrhage, uterine rupture, or PAS — the ureter is injured in 2–7% of emergency cesarean hysterectomies.[22]
- Parametrial dissection in PAS — deliberate dissection into the parametrium to gain vascular control or to excise placental tissue invading the parametrium places the ureter at the highest risk of any cesarean scenario.
- Repeat cesarean with cervical effacement and low-lying hysterotomy scar, where the ureters may track unusually close to the lateral angle of the hysterotomy.
GU Injuries During Cesarean — the Reconstructive Perspective
Bladder Injury
Incidence (see table above): 0.08–0.3% at primary cesarean; 0.6–1.0% at repeat cesarean; up to 29% in cesarean for PAS.[20][21][19]
Mechanisms:
- Peritoneal entry — sharp entry into a bladder adherent to the anterior abdominal wall (repeat CD). 20–50% of cesarean bladder injuries.
- Bladder flap dissection — sharp dissection outside the correct plane through densely scarred peritoneum.
- Hysterotomy — inadvertent low hysterotomy through a full bladder, or extension of the hysterotomy inferiorly into the bladder dome.
- Cesarean hysterectomy — dissection of the bladder off a scarred lower segment is the dominant mechanism here.
Recognition: clear fluid in the field, visible Foley balloon, hematuria, visible bladder mucosa. Intraoperative cystoscopy with indigo carmine or methylene blue retrograde fill is the highest-yield confirmation when injury is suspected.
Repair: recognized intraoperative cystotomy is repaired primarily in two layers (full-thickness absorbable; imbricating seromuscular) with postoperative catheter drainage for 7–14 days. A cystogram before catheter removal is reasonable for posterior, trigonal, or large injuries. Unrecognized bladder injury presents postoperatively as urinary ascites, ileus, urine leak from the incision, elevated drain creatinine, or — if the injury communicates with the uterine cavity or vagina — as a vesicouterine or vesicovaginal fistula (see below).
Ureteral Injury
Incidence: 0.03–0.09% at primary CD; 0.1–0.3% at repeat; 2–7% in emergency cesarean hysterectomy for PAS or hemorrhage.[22]
Mechanisms:
- Ligation / suture in the broad ligament during hemostatic control of a lateral extension — the dominant mechanism.
- Transection or thermal injury during cesarean hysterectomy at the cardinal ligament / uterine-artery pedicle.
- Kinking / devascularization from ureteral skeletonization during PAS dissection.
Location: the mid-ureter (pelvic brim to uterine artery crossing) is the typical site during hysterotomy extension; the distal intramural ureter is the typical site during cesarean hysterectomy.
Recognition: intraoperative — direct visualization of urine extravasation, a severed or clipped ureter, or loss of peristalsis. Postoperative — flank pain, fever, elevated creatinine, urinoma, urinary ascites, prolonged ileus, or flank hydronephrosis on imaging. Intraoperative cystoscopy with ureteral jet assessment after injecting IV indigo carmine / methylene blue is the highest-yield single maneuver for detection at the time of injury; its use is not standard at cesarean but should be considered whenever hemostatic sutures have been placed in the broad ligament or lateral angle.
Repair follows standard reconstructive principles — ureteroureterostomy for short mid-ureteral injuries, ureteroneocystostomy with psoas hitch or Boari flap for distal injuries, and Boari flap or ileal interposition for longer defects. Timing (immediate vs delayed with initial PCN and interval repair) depends on recognition timing, physiology, and operator comfort.
Vesicouterine Fistula (Youssef Syndrome)
A vesicouterine fistula (VUF) is a communication between the bladder and the uterine cavity. First described by Youssef in 1957,[26] the post-cesarean vesicouterine fistula has a characteristic clinical presentation — the Youssef triad:
- Menouria — cyclic hematuria during menstruation (menstrual blood drains into the bladder instead of the vagina)
- Amenorrhea (or reduced vaginal bleeding) — because menstrual blood preferentially drains through the fistula
- Urinary continence — because the trigone and bladder neck remain competent and the fistula sits above them
Cesarean delivery is the dominant etiology of VUF worldwide in high-cesarean-rate populations; a significant fraction of cases follow an apparently uncomplicated low-transverse cesarean where the bladder was not recognized as being injured at the time.[27][28] The low-transverse scar sits directly behind the posterior bladder wall; partial-thickness injury that later necroses or a suture placed through bladder into uterus are the two principal mechanisms.
Diagnosis is made by cystoscopy (direct visualization), VCUG (contrast passage into the uterine cavity), hysterosalpingography, or MRI. Spontaneous resolution with prolonged catheter drainage has been reported in small cases but is not reliable. Definitive management is surgical — transabdominal transvesical (O'Conor) or transabdominal extravesical repair with omental or peritoneal interposition. Transvaginal repair is technically difficult because the fistula sits high and anterior. Concurrent management of subsequent fertility desires must be discussed — the repair itself does not preclude future pregnancy, but a subsequent cesarean (mandatory) risks recurrence.
Uterine Niche / Isthmocele
Although not a GU injury per se, the cesarean scar niche is the anatomic substrate for several conditions that enter the reconstructive urologist's and urogynecologist's practice:
- Cesarean scar ectopic pregnancy — implantation within the niche, with 5–10% risk of uterine rupture or massive hemorrhage; increasingly managed by the reconstructive gynecologic surgeon.
- Placenta accreta spectrum in subsequent pregnancy (see below).
- Postmenstrual spotting and secondary dysmenorrhea — often misdiagnosed as other causes of abnormal uterine bleeding.
- Secondary infertility — hypothesized mechanism is retained menstrual blood in the niche impairing embryo transport or implantation.
Surgical repair of the niche — hysteroscopic resection of the superior edge (for fertility-preservation) or laparoscopic / vaginal excision and reapproximation of the defect — is a growing URPS domain.
Placenta Accreta Spectrum (PAS)
PAS is abnormal adherence of the placenta to the myometrium (accreta), into the myometrium (increta), or through the uterine serosa into adjacent structures (percreta). The dominant risk factor is prior cesarean with placenta previa in the current pregnancy — with risk rising from ~3% after one prior CD with previa to >40% after three or more prior CDs with previa.[19]
From the reconstructive urologist's perspective, PAS is the single obstetric scenario most likely to generate massive urinary-tract injury:
- Anterior percreta may invade the bladder wall directly. Attempting forcible separation is the classic mechanism of catastrophic bladder injury and massive hemorrhage.
- Parametrial percreta extends into the broad ligament, involving the ureter and the uterine vasculature.
- Planned cesarean hysterectomy with multidisciplinary team (MFM, GYN-ONC, urology, vascular, interventional radiology) is the contemporary standard of care in diagnosed PAS, often with preoperative ureteral stenting, prophylactic internal iliac balloon occlusion or embolization, and intraoperative urology availability for planned cystotomy and partial cystectomy when the bladder is involved.
Reconstructive urology is routinely consulted for:
- Intraoperative assistance at planned cesarean hysterectomy for PAS (ureteral stent placement, bladder dissection, intentional cystotomy and repair).
- Partial cystectomy with ureteral reimplantation when bladder invasion is confirmed.
- Postoperative management of fistulas, ureteral strictures, and bladder dysfunction following PAS surgery.
Implications for Reconstructive Repair Strategy
The original operation shapes every subsequent repair. A few patterns worth holding:
- Post-cesarean vesicouterine fistula is almost always supratrigonal, continent, and anatomically located where standard transvaginal VVF approaches do not reach — favoring transabdominal transvesical (O'Conor) repair with omental interposition.
- Post-cesarean vesicovaginal fistula (when present) tends to sit high on the anterior vaginal wall, near the bladder base — closer to the transvaginal approach but often reachable only with deliberate anterior vaginal wall flap mobilization.
- Post-cesarean ureteral injury is typically distal (either at the hysterotomy-extension level or at the cesarean-hysterectomy pedicle) — making ureteroneocystostomy ± psoas hitch the most common definitive repair.
- Post-cesarean retropubic fibrosis alters the anatomy of later Burch urethropexy, retropubic sling placement, and retropubic prostatectomy (for patients who subsequently undergo that operation at a later life stage).
- Repeat cesarean with incidentally recognized bladder injury repaired primarily in the operating room typically heals without sequelae; the disasters come from unrecognized injury.
Perioperative Care — Brief Orientation
Cesarean delivery operates under a well-codified enhanced-recovery framework. The ERAS Society has published 2025 updates in three parts — antenatal/preoperative, intraoperative, and postoperative — with strong recommendations for:[14][15][16]
- Single-dose first-generation cephalosporin within 60 minutes of incision
- Chlorhexidine-alcohol skin preparation; vaginal prep with povidone-iodine or chlorhexidine for laboring or ruptured-membrane patients
- Antiemetic and spinal-hypotension prophylaxis; normothermia; euvolemia; optimal uterotonic use; multimodal analgesia
- Early drinking, feeding, mobilization, and urinary-catheter removal
- Scheduled acetaminophen + NSAIDs; sparing opioid use
- Pneumatic compression for all patients; pharmacologic VTE prophylaxis (LMWH) for those with risk factors[17]
Urinary-catheter management is the ERAS element most relevant to reconstructive follow-up: early catheter removal reduces UTI and mobilization delay, but retention in the setting of neuraxial anesthesia is common and requires a structured re-catheterization pathway.
References
1. First and Second Stage Labor Management: ACOG Clinical Practice Guideline No. 8. Obstetrics and Gynecology. 2024;143(1):144–162. doi:10.1097/AOG.0000000000005447
2. Obstetric Care Consensus No. 1: Safe Prevention of the Primary Cesarean Delivery. Obstetrics and Gynecology. 2014;123(3):693–711. doi:10.1097/01.AOG.0000444441.04111.1d
3. Caughey AB, Cahill AG, Guise JM, Rouse DJ. Safe Prevention of the Primary Cesarean Delivery. American Journal of Obstetrics and Gynecology. 2014;210(3):179–193. doi:10.1016/j.ajog.2014.01.026
4. Mathai M, Hofmeyr GJ, Mathai NE. Abdominal Surgical Incisions for Caesarean Section. Cochrane Database of Systematic Reviews. 2013;(5):CD004453. doi:10.1002/14651858.CD004453.pub3
5. Gialdini C, Chamillard M, Diaz V, et al. Evidence-Based Surgical Procedures to Optimize Caesarean Outcomes: An Overview of Systematic Reviews. EClinicalMedicine. 2024;72:102632. doi:10.1016/j.eclinm.2024.102632
6. Hofmeyr JG, Novikova N, Mathai M, Shah A. Techniques for Cesarean Section. American Journal of Obstetrics and Gynecology. 2009;201(5):431–444. doi:10.1016/j.ajog.2009.03.018
7. Dominoni M, Torella M, Molitierno R, et al. Single- Versus Double-Layer Uterine Closure at the Time of Cesarean Delivery and Risk of Uterine Scar Niche: A Systematic Review and Meta-Analysis of Randomized Trials. Archives of Gynecology and Obstetrics. 2025. doi:10.1007/s00404-025-08151-y
8. Stegwee SI, van der Voet LF, Ben AJ, et al. Effect of Single- Versus Double-Layer Uterine Closure During Caesarean Section on Postmenstrual Spotting (2Close): Multicentre, Double-Blind, Randomised Controlled Superiority Trial. BJOG. 2021;128(5):866–878. doi:10.1111/1471-0528.16472
9. Verberkt C, Stegwee SI, Van der Voet LF, et al. Single-Layer vs Double-Layer Uterine Closure During Cesarean Delivery: 3-Year Follow-Up of a Randomized Controlled Trial (2Close Study). American Journal of Obstetrics and Gynecology. 2024;231(3):346.e1–346.e11. doi:10.1016/j.ajog.2023.12.032
10. CORONIS Collaborative Group, Abalos E, Addo V, et al. Caesarean Section Surgical Techniques (CORONIS): A Fractional, Factorial, Unmasked, Randomised Controlled Trial. Lancet. 2013;382(9888):234–248. doi:10.1016/S0140-6736(13)60441-9
11. Bujold E, Romero R. Uterine Closure After Cesarean Delivery: Surgical Technique, Biological Rationale, and Clinical Implications. American Journal of Obstetrics and Gynecology. 2026;233(6S):S82–S102. doi:10.1016/j.ajog.2025.10.007
12. Wang LH, Seow KM, Chen LR, Chen KH. The Health Impact of Surgical Techniques and Assistive Methods Used in Cesarean Deliveries: A Systematic Review. International Journal of Environmental Research and Public Health. 2020;17(18):6894. doi:10.3390/ijerph17186894
13. CORONIS Collaborative Group, Abalos E, Addo V, et al. Caesarean Section Surgical Techniques: 3-Year Follow-Up of the CORONIS Fractional, Factorial, Unmasked, Randomised Controlled Trial. Lancet. 2016;388(10039):62–72. doi:10.1016/S0140-6736(16)00204-X
14. Wilson RD, Monks DT, Sharawi N, et al. Guidelines for Antenatal and Preoperative Care in Cesarean Delivery: Enhanced Recovery After Surgery Society Recommendations (Part 1) — 2025 Update. American Journal of Obstetrics and Gynecology. 2025. doi:10.1016/j.ajog.2025.01.014
15. Caughey AB, Sultan P, Monks DT, et al. Guidelines for Intraoperative Care in Cesarean Delivery: Enhanced Recovery After Surgery Society Recommendations (Part 2) — 2025 Update. American Journal of Obstetrics and Gynecology. 2025. doi:10.1016/j.ajog.2025.02.040
16. Sultan P, Monks DT, Sharawi N, et al. Guidelines for Postoperative Care in Cesarean Delivery: Enhanced Recovery After Surgery Society Recommendations (Part 3) — 2025 Update. American Journal of Obstetrics and Gynecology. 2025. doi:10.1016/j.ajog.2025.01.038
17. Pacheco LD, Saade G, Metz TD. Society for Maternal-Fetal Medicine Consult Series #51: Thromboembolism Prophylaxis for Cesarean Delivery. American Journal of Obstetrics and Gynecology. 2020;223(2):B11–B17. doi:10.1016/j.ajog.2020.04.032
18. Sandall J, Tribe RM, Avery L, et al. Short-Term and Long-Term Effects of Caesarean Section on the Health of Women and Children. Lancet. 2018;392(10155):1349–1357. doi:10.1016/S0140-6736(18)31930-5
19. Silver RM, Landon MB, Rouse DJ, et al. Maternal Morbidity Associated With Multiple Repeat Cesarean Deliveries. Obstetrics and Gynecology. 2006;107(6):1226–1232. doi:10.1097/01.AOG.0000219750.79480.84
20. Phipps MG, Watabe B, Clemons JL, Weitzen S, Myers DL. Risk Factors for Bladder Injury During Cesarean Delivery. Obstetrics and Gynecology. 2005;105(1):156–160. doi:10.1097/01.AOG.0000149150.93552.78
21. Rajasekar D, Hall M. Urinary Tract Injuries During Obstetric Intervention. British Journal of Obstetrics and Gynaecology. 1997;104(6):731–734. doi:10.1111/j.1471-0528.1997.tb11986.x
22. Güngördük K, Asicioglu O, Celikkol O, Sudolmus S, Ark C. Iatrogenic Bladder Injuries During Caesarean Delivery: A Case Control Study. Journal of Obstetrics and Gynaecology. 2010;30(7):667–670. doi:10.3109/01443615.2010.494203
23. Antoine C, Pimentel VM, Timor-Tritsch IE, Mittal P, Bornstein E, Chervenak FA. Endometrium-Free Closure Technique for Hysterotomy Incision at Cesarean Delivery. American Journal of Obstetrics and Gynecology. 2026;233(6S). doi:10.1016/j.ajog.2025.07.041
24. Chelmow D, Rodriguez EJ, Sabatini MM. Suture Closure of Subcutaneous Fat and Wound Disruption After Cesarean Delivery: A Meta-Analysis. Obstetrics and Gynecology. 2004;103(5 Pt 1):974–980. doi:10.1097/01.AOG.0000124807.76451.47
25. Palacios-Jaraquemada JM, Nieto-Calvache AJ, Basanta NA, Aryananda RA, Martínez MA. Essential Anatomical Knowledge for Performing Routine and Complex Cesarean Delivery. American Journal of Obstetrics and Gynecology. 2026;233(6S):S368–S384. doi:10.1016/j.ajog.2025.08.012
26. Youssef AF. "Menouria" Following Lower Segment Cesarean Section: A Syndrome. American Journal of Obstetrics and Gynecology. 1957;73(4):759–767. doi:10.1016/0002-9378(57)90384-8
27. Tancer ML. Vesicouterine Fistula — A Review. Obstetrical and Gynecological Survey. 1986;41(12):743–753. doi:10.1097/00006254-198612000-00001
28. Hemal AK, Sharma SK, Mukherjee S. Ureteroscopy for Retrieval of Impacted Upper Ureteric Calculi; and Urological Management of Vesicouterine Fistulas in the Era of Minimally Invasive Surgery. BJU International. 2005;95(3):441–445.